Avian Influenza H7N9 Virus Adaptation to Human Hosts
- PMID: 34068495
- PMCID: PMC8150935
- DOI: 10.3390/v13050871
Avian Influenza H7N9 Virus Adaptation to Human Hosts
Abstract
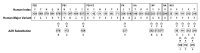
Avian influenza virus A (H7N9), after circulating in avian hosts for decades, was identified as a human pathogen in 2013. Herein, amino acid substitutions possibly essential for human adaptation were identified by comparing the 4706 aligned overlapping nonamer position sequences (1-9, 2-10, etc.) of the reported 2014 and 2017 avian and human H7N9 datasets. The initial set of virus sequences (as of year 2014) exhibited a total of 109 avian-to-human (A2H) signature amino acid substitutions. Each represented the most prevalent substitution at a given avian virus nonamer position that was selectively adapted as the corresponding index (most prevalent sequence) of the human viruses. The majority of these avian substitutions were long-standing in the evolution of H7N9, and only 17 were first detected in 2013 as possibly essential for the initial human adaptation. Strikingly, continued evolution of the avian H7N9 virus has resulted in avian and human protein sequences that are almost identical. This rapid and continued adaptation of the avian H7N9 virus to the human host, with near identity of the avian and human viruses, is associated with increased human infection and a predicted greater risk of human-to-human transmission.
Keywords: H7N9; adaptation; avian viruses; diversity; host; human viruses; influenza virus; motifs; surveillance; zoonosis.
Conflict of interest statement
We declare that we have no competing interests in this research.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

