All-Trans Retinoic Acid Increases DRP1 Levels and Promotes Mitochondrial Fission
- PMID: 34068960
- PMCID: PMC8156392
- DOI: 10.3390/cells10051202
All-Trans Retinoic Acid Increases DRP1 Levels and Promotes Mitochondrial Fission
Abstract
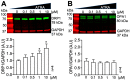
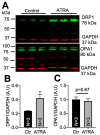
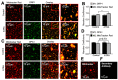
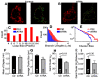
In the heart, mitochondrial homeostasis is critical for sustaining normal function and optimal responses to metabolic and environmental stressors. Mitochondrial fusion and fission are thought to be necessary for maintaining a robust population of mitochondria, and disruptions in mitochondrial fission and/or fusion can lead to cellular dysfunction. The dynamin-related protein (DRP1) is an important mediator of mitochondrial fission. In this study, we investigated the direct effects of the micronutrient retinoid all-trans retinoic acid (ATRA) on the mitochondrial structure in vivo and in vitro using Western blot, confocal, and transmission electron microscopy, as well as mitochondrial network quantification using stochastic modeling. Our results showed that ATRA increases DRP1 protein levels, increases the localization of DRP1 to mitochondria in isolated mitochondrial preparations. Our results also suggested that ATRA remodels the mitochondrial ultrastructure where the mitochondrial area and perimeter were decreased and the circularity was increased. Microscopically, mitochondrial network remodeling is driven by an increased rate of fission over fusion events in ATRA, as suggested by our numerical modeling. In conclusion, ATRA results in a pharmacologically mediated increase in the DRP1 protein. It also results in the modulation of cardiac mitochondria by promoting fission events, altering the mitochondrial network, and modifying the ultrastructure of mitochondria in the heart.
Keywords: DRP1; all-trans retinoic acid; mitochondrial fission; mitochondrial fusion; mitochondrial network.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

