Efficacy and Safety of [225Ac]Ac-PSMA-617 Augmented [177Lu]Lu-PSMA-617 Radioligand Therapy in Patients with Highly Advanced mCRPC with Poor Prognosis
- PMID: 34069003
- PMCID: PMC8156464
- DOI: 10.3390/pharmaceutics13050722
Efficacy and Safety of [225Ac]Ac-PSMA-617 Augmented [177Lu]Lu-PSMA-617 Radioligand Therapy in Patients with Highly Advanced mCRPC with Poor Prognosis
Abstract
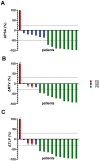
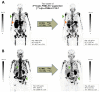
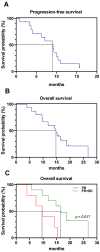
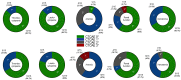
The use of 225Ac in prostate-specific membrane antigen (PSMA)-targeted radioligand therapy (RLT), either as monotherapy or in combination with 177Lu, is a promising therapy approach in patients with metastatic castration-resistant prostate carcinoma (mCRPC). In this study, we report the efficacy and safety of [225Ac]Ac-PSMA-617 augmented [177Lu]Lu-PSMA-617 RLT in 177Lu-naive mCRPC patients (n = 15) with poor prognosis (presence of visceral metastases, high total tumor burden with diffuse bone metastases or a short PSA doubling time of <2 months). Biochemical (by PSA serum value) and molecular imaging response (by [68Ga]Ga-PSMA-11 PET/CT) was assessed after two cycles of [177Lu]Lu-PSMA-617 RLT, with at least one [225Ac]Ac-PSMA-617 augmentation. In addition, PSA-based progression-free survival (PSA-PFS), overall survival (OS) and toxicity (according to CTCAE) were analyzed. We observed a biochemical- and molecular imaging-based partial remission in 53.3% (8/15) and 66.7% (10/15) of patients, respectively. The median PSA-PFS and OS was 9.1 and 14.8 months, respectively. No serious acute adverse events were recorded. Two out of fifteen patients experienced grade 3 anemia. No other grade 3/4 toxicities were observed. RLT-related xerostomia (grade 1/2) was recorded in 2/15 patients. Our data showed a high clinical efficacy with a favorable side effects profile of [225Ac]Ac-PSMA-617 augmented [177Lu]Lu-PSMA-617 RLT in this highly challenging patient cohort.
Keywords: 225Ac and 177Lu; PSMA radioligand therapy; biochemical response; efficacy; metastatic castration-resistant prostate cancer; molecular imaging response; toxicity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Efficacy and safety of 225Ac-PSMA-617 targeted alpha therapy in metastatic castration-resistant Prostate Cancer patients.Theranostics. 2020 Jul 23;10(20):9364-9377. doi: 10.7150/thno.48107. eCollection 2020. Theranostics. 2020. PMID: 32802197 Free PMC article.
-
Molecular imaging and biochemical response assessment after a single cycle of [225Ac]Ac-PSMA-617/[177Lu]Lu-PSMA-617 tandem therapy in mCRPC patients who have progressed on [177Lu]Lu-PSMA-617 monotherapy.Theranostics. 2021 Feb 16;11(9):4050-4060. doi: 10.7150/thno.56211. eCollection 2021. Theranostics. 2021. PMID: 33754047 Free PMC article.
-
Activity and Adverse Events of Actinium-225-PSMA-617 in Advanced Metastatic Castration-resistant Prostate Cancer After Failure of Lutetium-177-PSMA.Eur Urol. 2021 Mar;79(3):343-350. doi: 10.1016/j.eururo.2020.11.013. Epub 2020 Dec 5. Eur Urol. 2021. PMID: 33293081
-
Targeted Alpha Therapy: Exploring the Clinical Insights into [225Ac]Ac-PSMA and Its Relevance Compared with [177Lu]Lu-PSMA in Advanced Prostate Cancer Management.Pharmaceuticals (Basel). 2025 Aug 18;18(8):1215. doi: 10.3390/ph18081215. Pharmaceuticals (Basel). 2025. PMID: 40872606 Free PMC article. Review.
-
Hematologic toxicity profile and efficacy of [225Ac]Ac-PSMA-617 α-radioligand therapy of patients with extensive skeletal metastases of castration-resistant prostate cancer.Eur J Nucl Med Mol Imaging. 2022 Aug;49(10):3581-3592. doi: 10.1007/s00259-022-05778-w. Epub 2022 Apr 6. Eur J Nucl Med Mol Imaging. 2022. PMID: 35384462 Review.
Cited by
-
Tandem Isotope Therapy with 225Ac- and 177Lu-PSMA-617 in a Murine Model of Prostate Cancer.J Nucl Med. 2023 Nov;64(11):1772-1778. doi: 10.2967/jnumed.123.265433. Epub 2023 Oct 5. J Nucl Med. 2023. PMID: 37797974 Free PMC article.
-
Emerging Role of Nuclear Medicine in Prostate Cancer: Current State and Future Perspectives.Cancers (Basel). 2023 Sep 27;15(19):4746. doi: 10.3390/cancers15194746. Cancers (Basel). 2023. PMID: 37835440 Free PMC article. Review.
-
Radiopharmaceutical Treatments for Cancer Therapy, Radionuclides Characteristics, Applications, and Challenges.Molecules. 2022 Aug 16;27(16):5231. doi: 10.3390/molecules27165231. Molecules. 2022. PMID: 36014472 Free PMC article. Review.
-
PSMA Theranostics: Science and Practice.Cancers (Basel). 2021 Aug 2;13(15):3904. doi: 10.3390/cancers13153904. Cancers (Basel). 2021. PMID: 34359805 Free PMC article. Review.
-
PSMA radioligand therapy rechallenging: expanding the therapeutic possibilities in metastatic castration resistant prostate cancer.Eur J Nucl Med Mol Imaging. 2024 Nov;51(13):4163-4164. doi: 10.1007/s00259-024-06870-z. Eur J Nucl Med Mol Imaging. 2024. PMID: 39096364 No abstract available.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

