Transferrin Modified GSH Sensitive Hyaluronic Acid Derivative Micelle to Deliver HSP90 Inhibitors to Enhance the Therapeutic Efficacy of Brain Cancers
- PMID: 34069106
- PMCID: PMC8156315
- DOI: 10.3390/cancers13102375
Transferrin Modified GSH Sensitive Hyaluronic Acid Derivative Micelle to Deliver HSP90 Inhibitors to Enhance the Therapeutic Efficacy of Brain Cancers
Abstract
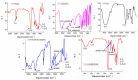
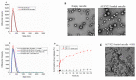
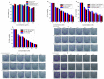
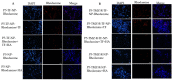
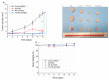
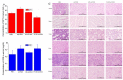
Herein, GSH-sensitive hyaluronic acid-poly(lactic-co-glycolic acid) (HA-SS-PLGA) was synthesized. Surface modification of PLGA with hyaluronic acid produced a highly stable micelle at physiological pH while a micelle was destabilized at a higher GSH level. Fluorescence microscopy results showed that rhodamine-encapsulated micelle was taken up by brain cancer cells, while competitive inhibition was observed in the presence of free HA and free transferrin. In vitro cytotoxicity results revealed that transferrin-targeted nanoformulated AUY922 (TF-NP-AUY922) shows higher cytotoxicity than either free AUY922 or non-targeted AUY922-loaded micelles (NP-AUY922). In comparison to the control groups, free AUY922, TF-NP-AUY922 or NP-AUY922 treatment revealed the upregulation of HSP70, while the expression of HSP90 client proteins was simultaneously depleted. In addition, the treatment group induced caspase-dependent PARP cleavage and the upregulation of p53 expression, which plays a key role in apoptosis of brain cancer cells. In vivo and ex vivo biodistribution studies showed that cypate-loaded micelle was taken up and accumulated in the tumor regions. Furthermore, in vivo therapeutic efficacy studies revealed that the AUY922-loaded micelle significantly suppressed tumor growth in comparison to the free AUY922, or control groups using tumor-bearing NOD-SCID mice. Moreover, biochemical index and histological analysis revealed synthesized micelle does not show any significant cytotoxicity to the selected major organs. Overall, a synthesized micelle is the best carrier for AUY922 to enhance the therapeutic efficiency of brain cancer.
Keywords: GSH-sensitive micelle; brain cancer; hyaluronic acid; poly(lactic-co-glycolic) acid.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures






Similar articles
-
pH- and GSH-Sensitive Hyaluronic Acid-MP Conjugate Micelles for Intracellular Delivery of Doxorubicin to Colon Cancer Cells and Cancer Stem Cells.Biomacromolecules. 2018 Sep 10;19(9):3725-3737. doi: 10.1021/acs.biomac.8b00856. Epub 2018 Aug 9. Biomacromolecules. 2018. PMID: 30044910
-
Combination Delivery of Alpha-Tocopheryl Succinate and Curcumin Using a GSH-Sensitive Micelle (PAH-SS-PLGA) to Treat Pancreatic Cancer.Pharmaceutics. 2020 Aug 16;12(8):778. doi: 10.3390/pharmaceutics12080778. Pharmaceutics. 2020. PMID: 32824299 Free PMC article.
-
Multifunctional Hyaluronic Acid-Decorated Redox-Responsive Magnetic Complex Micelle for Targeted Drug Delivery with Enhanced Antitumor Efficiency and Anti-Cell-Migration Activity.J Biomed Nanotechnol. 2018 Mar 1;14(3):477-495. doi: 10.1166/jbn.2018.2541. J Biomed Nanotechnol. 2018. PMID: 29663921
-
Targeting HSP90 with the small molecule inhibitor AUY922 (luminespib) as a treatment strategy against hepatocellular carcinoma.Int J Cancer. 2019 May 15;144(10):2613-2624. doi: 10.1002/ijc.31963. Epub 2018 Dec 24. Int J Cancer. 2019. PMID: 30488605
-
Poly[lactic-co-(glycolic acid)]-grafted hyaluronic acid copolymer micelle nanoparticles for target-specific delivery of doxorubicin.Macromol Biosci. 2009 Apr 8;9(4):336-42. doi: 10.1002/mabi.200800229. Macromol Biosci. 2009. PMID: 19006195
Cited by
-
Antioxidants in brain tumors: current therapeutic significance and future prospects.Mol Cancer. 2022 Oct 28;21(1):204. doi: 10.1186/s12943-022-01668-9. Mol Cancer. 2022. PMID: 36307808 Free PMC article. Review.
-
Light-Decomposable Polymeric Micelles with Hypoxia-Enhanced Phototherapeutic Efficacy for Combating Metastatic Breast Cancer.Pharmaceutics. 2022 Jan 21;14(2):253. doi: 10.3390/pharmaceutics14020253. Pharmaceutics. 2022. PMID: 35213986 Free PMC article.
-
Targeting Brain Drug Delivery with Macromolecules Through Receptor-Mediated Transcytosis.Pharmaceutics. 2025 Jan 15;17(1):109. doi: 10.3390/pharmaceutics17010109. Pharmaceutics. 2025. PMID: 39861756 Free PMC article. Review.
-
Hyaluronic Acid within Self-Assembling Nanoparticles: Endless Possibilities for Targeted Cancer Therapy.Nanomaterials (Basel). 2022 Aug 18;12(16):2851. doi: 10.3390/nano12162851. Nanomaterials (Basel). 2022. PMID: 36014715 Free PMC article. Review.
References
-
- Rock K., McArdle O., Forde P., Dunne M., Fitzpatrick D., O’Neill B., Faul C. A clinical review of treatment outcomes in glioblastoma multiforme--the validation in a non-trial population of the results of a randomised Phase III clinical trial: Has a more radical approach improved survival? Br. J. Radiol. 2012;85:e729–e733. doi: 10.1259/bjr/83796755. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

