New Cardenolides from Biotransformation of Gitoxigenin by the Endophytic Fungus Alternaria eureka 1E1BL1: Characterization and Cytotoxic Activities
- PMID: 34069653
- PMCID: PMC8161373
- DOI: 10.3390/molecules26103030
New Cardenolides from Biotransformation of Gitoxigenin by the Endophytic Fungus Alternaria eureka 1E1BL1: Characterization and Cytotoxic Activities
Abstract
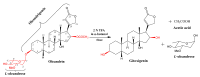
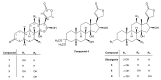
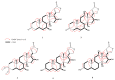
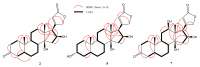
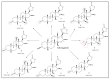
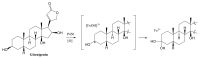
Microbial biotransformation is an important tool in drug discovery and for metabolism studies. To expand our bioactive natural product library via modification and to identify possible mammalian metabolites, a cytotoxic cardenolide (gitoxigenin) was biotransformed using the endophytic fungus Alternaria eureka 1E1BL1. Initially, oleandrin was isolated from the dried leaves of Nerium oleander L. and subjected to an acid-catalysed hydrolysis to obtain the substrate gitoxigenin (yield; ~25%). After 21 days of incubation, five new cardenolides 1, 3, 4, 6, and 8 and three previously- identified compounds 2, 5 and 7 were isolated using chromatographic methods. Structural elucidations were accomplished through 1D/2D NMR, HR-ESI-MS and FT-IR analysis. A. eureka catalyzed oxygenation, oxidation, epimerization and dimethyl acetal formation reactions on the substrate. Cytotoxicity of the metabolites were evaluated using MTT cell viability method, whereas doxorubicin and oleandrin were used as positive controls. Biotransformation products displayed less cytotoxicity than the substrate. The new metabolite 8 exhibited the highest activity with IC50 values of 8.25, 1.95 and 3.4 µM against A549, PANC-1 and MIA PaCa-2 cells, respectively, without causing toxicity on healthy cell lines (MRC-5 and HEK-293) up to concentration of 10 µM. Our results suggest that A. eureka is an effective biocatalyst for modifying cardenolide-type secondary metabolites.
Keywords: Alternaria eureka 1E1BL1; Nerium oleander L.; biotransformation; cardenolides; cytotoxicity; endophytic fungus; gitoxigenin; oleandrin.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Biotransformation of cyclocanthogenol by the endophytic fungus Alternaria eureka 1E1BL1.Phytochemistry. 2018 Jul;151:91-98. doi: 10.1016/j.phytochem.2018.04.006. Epub 2018 Apr 17. Phytochemistry. 2018. PMID: 29677643
-
Cardenolides from the leaves of Nerium oleander.Fitoterapia. 2018 Jun;127:293-300. doi: 10.1016/j.fitote.2018.03.004. Epub 2018 Mar 11. Fitoterapia. 2018. PMID: 29540313
-
Biotransformation of Neoruscogenin by the Endophytic Fungus Alternaria eureka.J Nat Prod. 2018 Jun 22;81(6):1357-1367. doi: 10.1021/acs.jnatprod.7b00898. Epub 2018 Jun 12. J Nat Prod. 2018. PMID: 29893560
-
Oleandrin: A bioactive phytochemical and potential cancer killer via multiple cellular signaling pathways.Food Chem Toxicol. 2020 Sep;143:111570. doi: 10.1016/j.fct.2020.111570. Epub 2020 Jul 5. Food Chem Toxicol. 2020. PMID: 32640345 Review.
-
Cardenolides from the Apocynaceae family and their anticancer activity.Fitoterapia. 2016 Jul;112:74-84. doi: 10.1016/j.fitote.2016.04.023. Epub 2016 May 7. Fitoterapia. 2016. PMID: 27167183 Review.
Cited by
-
Luminescent Properties of β-(hydroxyaryl)-butenolides and Fluorescence Quenching in Water.J Fluoresc. 2025 Feb;35(2):905-920. doi: 10.1007/s10895-023-03546-z. Epub 2024 Jan 9. J Fluoresc. 2025. PMID: 38193954
-
Anabasis setifera leaf extract from arid habitat: A treasure trove of bioactive phytochemicals with potent antimicrobial, anticancer, and antioxidant properties.PLoS One. 2024 Oct 25;19(10):e0310298. doi: 10.1371/journal.pone.0310298. eCollection 2024. PLoS One. 2024. PMID: 39453934 Free PMC article.
-
Biotransformation of selected secondary metabolites by Alternaria species and the pharmaceutical, food and agricultural application of biotransformation products.Nat Prod Bioprospect. 2024 Aug 19;14(1):46. doi: 10.1007/s13659-024-00469-5. Nat Prod Bioprospect. 2024. PMID: 39158793 Free PMC article. Review.
-
Global regulatory factor VeA upregulates the production of antitumor substances in endophytic Fusarium solani.Antonie Van Leeuwenhoek. 2022 Aug;115(8):1085-1100. doi: 10.1007/s10482-022-01753-5. Epub 2022 Jul 5. Antonie Van Leeuwenhoek. 2022. PMID: 35789442
-
Neuroprotective metabolites via fungal biotransformation of a novel sapogenin, cyclocephagenol.Sci Rep. 2022 Nov 2;12(1):18481. doi: 10.1038/s41598-022-22799-5. Sci Rep. 2022. PMID: 36323752 Free PMC article.
References
-
- Li H., Gilchrist C.L.M., Phan C.-S., Lacey H.J., Vuong D., Moggach S.A., Lacey E., Piggott A.M., Chooi Y.-H. Biosynthesis of a new benzazepine alkaloid nanangelenin A from Aspergillus nanangensis Involves an unusual l-kynurenine-incorporating NRPS catalyzing regioselective lactamization. J. Am. Chem. Soc. 2020;142:7145–7152. doi: 10.1021/jacs.0c01605. - DOI - PubMed
-
- Monagas M., Urpi-Sarda M., Sánchez-Patán F., Llorach R., Garrido I., Gómez-Cordovés C., Andres-Lacueva C., Bartolomé B. Insights into the metabolism and microbial biotransformation of dietary flavan-3-ols and the bioactivity of their metabolites. Food Funct. 2010;1:233–253. doi: 10.1039/c0fo00132e. - DOI - PubMed
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

