Accessory Subunits of the Matrix Arm of Mitochondrial Complex I with a Focus on Subunit NDUFS4 and Its Role in Complex I Function and Assembly
- PMID: 34069703
- PMCID: PMC8161149
- DOI: 10.3390/life11050455
Accessory Subunits of the Matrix Arm of Mitochondrial Complex I with a Focus on Subunit NDUFS4 and Its Role in Complex I Function and Assembly
Abstract
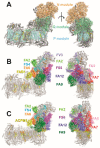
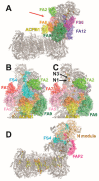
NADH:ubiquinone-oxidoreductase (complex I) is the largest membrane protein complex of the respiratory chain. Complex I couples electron transfer to vectorial proton translocation across the inner mitochondrial membrane. The L shaped structure of complex I is divided into a membrane arm and a matrix arm. Fourteen central subunits are conserved throughout species, while some 30 accessory subunits are typically found in eukaryotes. Complex I dysfunction is associated with mutations in the nuclear and mitochondrial genome, resulting in a broad spectrum of neuromuscular and neurodegenerative diseases. Accessory subunit NDUFS4 in the matrix arm is a hot spot for mutations causing Leigh or Leigh-like syndrome. In this review, we focus on accessory subunits of the matrix arm and discuss recent reports on the function of accessory subunit NDUFS4 and its interplay with NDUFS6, NDUFA12, and assembly factor NDUFAF2 in complex I assembly.
Keywords: Leigh syndrome; NADH dehydrogenase; assembly factor; mitochondrial disease; oxidative phosphorylation; respiratory chain.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
NDUFS4 deletion triggers loss of NDUFA12 in Ndufs4-/- mice and Leigh syndrome patients: A stabilizing role for NDUFAF2.Biochim Biophys Acta Bioenerg. 2020 Aug 1;1861(8):148213. doi: 10.1016/j.bbabio.2020.148213. Epub 2020 Apr 23. Biochim Biophys Acta Bioenerg. 2020. PMID: 32335026
-
Structural insights into respiratory complex I deficiency and assembly from the mitochondrial disease-related ndufs4-/- mouse.EMBO J. 2024 Jan;43(2):225-249. doi: 10.1038/s44318-023-00001-4. Epub 2024 Jan 2. EMBO J. 2024. PMID: 38177503 Free PMC article.
-
Nuclear Gene-Encoded Leigh Syndrome Spectrum Overview.2015 Oct 1 [updated 2025 May 1]. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2025. 2015 Oct 1 [updated 2025 May 1]. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2025. PMID: 26425749 Free Books & Documents. Review.
-
Accessory subunit NUYM (NDUFS4) is required for stability of the electron input module and activity of mitochondrial complex I.Biochim Biophys Acta Bioenerg. 2017 Feb;1858(2):175-181. doi: 10.1016/j.bbabio.2016.11.010. Epub 2016 Nov 19. Biochim Biophys Acta Bioenerg. 2017. PMID: 27871794
-
Ndufs4 knockout mouse models of Leigh syndrome: pathophysiology and intervention.Brain. 2022 Mar 29;145(1):45-63. doi: 10.1093/brain/awab426. Brain. 2022. PMID: 34849584 Free PMC article. Review.
Cited by
-
Proton-Translocating NADH-Ubiquinone Oxidoreductase: Interaction with Artificial Electron Acceptors, Inhibitors, and Potential Medicines.Int J Mol Sci. 2024 Dec 14;25(24):13421. doi: 10.3390/ijms252413421. Int J Mol Sci. 2024. PMID: 39769185 Free PMC article. Review.
-
Enhanced Anticancer Activity of 7MeERT over Ertredin: A Comparative Study on Cancer Cell Proliferation and NDUFA12 Binding.Biomolecules. 2024 Sep 23;14(9):1197. doi: 10.3390/biom14091197. Biomolecules. 2024. PMID: 39334963 Free PMC article.
-
Homozygous Microdeletion Involving Exon 1 of ERCC8 and NDUFAF2 With Uniparental Isodisomy of Chromosome 5.Mol Genet Genomic Med. 2024 Dec;12(12):e70037. doi: 10.1002/mgg3.70037. Mol Genet Genomic Med. 2024. PMID: 39621529 Free PMC article.
-
Insights into Molecular Mechanisms of Anticancer Activity of Juniperus communis Essential Oil in HeLa and HCT 116 Cells.Plants (Basel). 2024 Aug 23;13(17):2351. doi: 10.3390/plants13172351. Plants (Basel). 2024. PMID: 39273835 Free PMC article.
-
Alternative splicing expands the clinical spectrum of NDUFS6-related mitochondrial disorders.Genet Med. 2024 Jun;26(6):101117. doi: 10.1016/j.gim.2024.101117. Epub 2024 Mar 6. Genet Med. 2024. PMID: 38459834 Free PMC article.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

