Comparative Structural Study of Three Tetrahalophthalic Anhydrides: Recognition of X···O(anhydride) Halogen Bond and πh···O(anhydride) Interaction
- PMID: 34071107
- PMCID: PMC8197102
- DOI: 10.3390/molecules26113119
Comparative Structural Study of Three Tetrahalophthalic Anhydrides: Recognition of X···O(anhydride) Halogen Bond and πh···O(anhydride) Interaction
Abstract
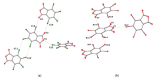
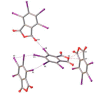
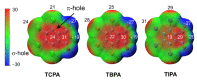
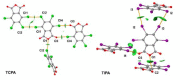
Structures of three tetrahalophthalic anhydrides (TXPA: halogen = Cl (TCPA), Br (TBPA), I (TIPA)) were studied by X-ray diffraction, and several types of halogen bonds (HaB) and lone pair···π-hole (lp···πh) contacts were revealed in their structures. HaBs involving the central oxygen atom of anhydride group (further X···O(anhydride) were recognized in the structures of TCPA and TBPA. In contrast, for the O(anhydride) atom of TIPA, only interactions with the π system (π-hole) of the anhydride ring (further lp(O)···πh) were observed. Computational studies by a number of theoretical methods (molecular electrostatic potentials, the quantum theory of atoms in molecules, the independent gradient model, natural bond orbital analyses, the electron density difference, and symmetry-adapted perturbation theory) demonstrated that the X···O(anhydride) contacts in TCPA and TBPA and lp(O)···πh in TIPA are caused by the packing effect. The supramolecular architecture of isostructural TCPA and TBPA was mainly affected by X···O(acyl) and X···X HaBs, and, for TIPA, the main contribution provided I···I HaBs.
Keywords: X-ray diffraction studies; computational studies; halogen bond; noncovalent interactions; tetrahalophthalic anhydrides.
Conflict of interest statement
The authors declare no competing interests.
Figures




Similar articles
-
Diverse Self-Assembled Molecular Architectures Promoted by C-H···O and C-H···Cl Hydrogen Bonds in a Triad of α-Diketone, α-Ketoimine, and an Imidorhenium Complex: A Unified Analysis Based on XRD, NEDA, SAPT, QTAIM, and IBSI Studies.ACS Omega. 2024 Nov 2;9(45):45518-45536. doi: 10.1021/acsomega.4c07702. eCollection 2024 Nov 12. ACS Omega. 2024. PMID: 39554419 Free PMC article.
-
Chalcogen- and halogen-bonds involving SX2 (X = F, Cl, and Br) with formaldehyde.J Mol Model. 2016 Jul;22(7):167. doi: 10.1007/s00894-016-3037-6. Epub 2016 Jun 24. J Mol Model. 2016. PMID: 27342252
-
The enhancing effects of group V σ-hole interactions on the F···O halogen bond.Phys Chem Chem Phys. 2014 Sep 28;16(36):19282-9. doi: 10.1039/c4cp02430c. Phys Chem Chem Phys. 2014. PMID: 25099757
-
Halogen-π Interactions between Benzene and X2/CX4 (X = Cl, Br): Assessment of Various Density Functionals with Respect to CCSD(T).J Phys Chem A. 2016 Nov 23;120(46):9305-9314. doi: 10.1021/acs.jpca.6b09395. Epub 2016 Nov 14. J Phys Chem A. 2016. PMID: 27802060
-
Theoretical ab Initio Study on Cooperativity Effects between Nitro π-hole and Halogen Bonding Interactions.Chemphyschem. 2019 May 3;20(9):1135-1144. doi: 10.1002/cphc.201900142. Epub 2019 Apr 10. Chemphyschem. 2019. PMID: 30884031
References
-
- Isakov D., Vasilev S., Gomes E.d.M., Almeida B., Shur V.Y., Kholkin A.L. Probing Ferroelectric Behaviour in Charge-Transfer Organic Meta-Nitroaniline. Appl. Phys. Lett. 2016;109:162903. doi: 10.1063/1.4965710. - DOI
-
- Miller J.S., Epstein A.J., Reiff W.M. Ferromagnetic Molecular Charge-Transfer Complexes. Chem. Rev. 1988;88:201–220. doi: 10.1021/cr00083a010. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

