Multiplex Autoantibody Detection in Patients with Autoimmune Polyglandular Syndromes
- PMID: 34071130
- PMCID: PMC8197071
- DOI: 10.3390/ijms22115502
Multiplex Autoantibody Detection in Patients with Autoimmune Polyglandular Syndromes
Abstract
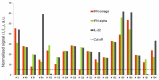
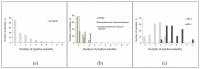
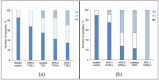
The diagnosis of autoimmune polyglandular syndrome (APS) types 1/2 is difficult due to their rarity and nonspecific clinical manifestations. APS-1 development can be identified with assays for autoantibodies against cytokines, and APS-2 development with organ-specific antibodies. In this study, a microarray-based multiplex assay was proposed for simultaneous detection of both organ-specific (anti-21-OH, anti-GAD-65, anti-IA2, anti-ICA, anti-TG, and anti-TPO) and APS-1-specific (anti-IFN-ω, anti-IFN-α-2a, and anti-IL-22) autoantibodies. Herein, 206 serum samples from adult patients with APS-1, APS-2, isolated autoimmune endocrine pathologies or non-autoimmune endocrine pathologies and from healthy donors were analyzed. The prevalence of autoantibodies differed among the groups of healthy donors and patients with non-, mono- and multi-endocrine diseases. APS-1 patients were characterized by the presence of at least two specific autoantibodies (specificity 99.5%, sensitivity 100%). Furthermore, in 16 of the 18 patients, the APS-1 assay revealed triple positivity for autoantibodies against IFN-ω, IFN-α-2a and IL-22 (specificity 100%, sensitivity 88.9%). No anti-cytokine autoantibodies were found in the group of patients with non-APS-1 polyendocrine autoimmunity. The accuracy of the microarray-based assay compared to ELISA for organ-specific autoantibodies was 88.8-97.6%. This multiplex assay can be part of the strategy for diagnosing and predicting the development of APS.
Keywords: autoantibodies; autoimmune polyglandular syndrome; microarray; multiplex assay.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Singh G., Jialal I. StatPearls [Internet] StatPearls Publishing; Treasure Island, FL, USA: 2020. Polyglandular Autoimmune Syndrome Type II.
-
- Puel A., Döffinger R., Natividad A., Chrabieh M., Barcenas-Morales G., Picard C., Cobat A., Ouachée-Chardin M., Toulon A., Bustamante J., et al. Autoantibodies against IL-17A, IL-17F, and IL-22 in patients with chronic mucocutaneous candidiasis and autoimmune polyendocrine syndrome type I. J. Exp. Med. 2010;207:291–297. doi: 10.1084/jem.20091983. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

