Short-Term Ex Vivo Culture of CTCs from Advance Breast Cancer Patients: Clinical Implications
- PMID: 34071445
- PMCID: PMC8198105
- DOI: 10.3390/cancers13112668
Short-Term Ex Vivo Culture of CTCs from Advance Breast Cancer Patients: Clinical Implications
Abstract
Background: Circulating tumor cells (CTC) have relevance as prognostic markers in breast cancer. However, the functional properties of CTCs or their molecular characterization have not been well-studied. Experimental models indicate that only a few cells can survive in the circulation and eventually metastasize. Thus, it is essential to identify these surviving cells capable of forming such metastases.
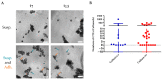
Methods: We isolated viable CTCs from 50 peripheral blood samples obtained from 35 patients with advanced metastatic breast cancer using RosetteSepTM for ex vivo culture. The CTCs were seeded and monitored on plates under low adherence conditions and with media supplemented with growth factors and Nanoemulsions. Phenotypic analysis was performed by immunofluorescence and gene expression analysis using RT-PCR and CTCs counting by the Cellsearch® system.
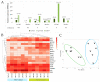
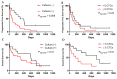
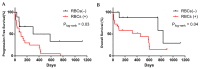
Results: We found that in 75% of samples the CTC cultures lasted more than 23 days, predicting a shorter Progression-Free Survival in these patients, independently of having ≥5 CTC by Cellsearch®. We also observed that CTCs before and after culture showed a different gene expression profile.
Conclusions: the cultivability of CTCs is a predictive factor. Furthermore, the subset of cells capable of growing ex vivo show stem or mesenchymal features and may represent the CTC population with metastatic potential in vivo.
Keywords: CTC; breast cancer; cell culture; liquid biopsy.
Conflict of interest statement
R.L.-L. reports grants and personal fees from Roche, Merck, AstraZeneca, Bayer, Pharmamar, Leo, and personal fees and non-financial support from Bristol-Myers Squibb and Novartis, outside of the submitted work. The other authors declare no conflict of interest. A patent entitled “Nanosystems for cellular proliferation” (PCT/EP2018/079214) has been deposited describing the effect of these Nanoemulsions in breast cancer cells.
Figures


References
-
- Bidard F.-C., Peeters D.J., Fehm T., Nolé F., Gisbert-Criado R., Mavroudis D., Grisanti S., Generali D., Garcia-Saenz J.A., Stebbing J., et al. Clinical validity of circulating tumour cells in patients with metastatic breast cancer: A pooled analysis of individual patient data. Lancet Oncol. 2014;15:406–414. doi: 10.1016/S1470-2045(14)70069-5. - DOI - PubMed
-
- De Bono J.S., Scher H.I., Montgomery R.B., Parker C., Miller M.C., Tissing H., Doyle G.V., Terstappen L.W., Pienta K., Raghavan D. Circulating Tumor Cells Predict Survival Benefit from Treatment in Metastatic Castration-Resistant Prostate Cancer. Clin. Cancer Res. 2008;14:6302–6309. doi: 10.1158/1078-0432.CCR-08-0872. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

