Flow-Through Electrochemical Biosensor for the Detection of Listeria monocytogenes Using Oligonucleotides
- PMID: 34071528
- PMCID: PMC8198859
- DOI: 10.3390/s21113754
Flow-Through Electrochemical Biosensor for the Detection of Listeria monocytogenes Using Oligonucleotides
Abstract
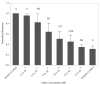
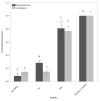
Consumption of food contaminated by Listeria monocytogenes can result in Listeriosis, an illness with hospitalization rates of 94% and mortality rates up to 30%. As a result, U.S. regulatory agencies governing food safety retain zero-tolerance policies for L. monocytogenes. However, detection at such low concentrations often requires strategies such as increasing sample size or culture enrichment. A novel flow-through immunoelectrochemical biosensor has been developed for Escherichia coli O157:H7 detection in 1 L volumes without enrichment. The current work further augments this biosensor's capabilities to (1) include detection of L. monocytogenes and (2) accommodate genetic detection to help overcome limitations based upon antibody availability and address specificity errors in phenotypic assays. Herein, the conjugation scheme for oligo attachment and the conditions necessary for genetic detection are laid forth while results of the present study demonstrate the sensor's ability to distinguish L. monocytogenes DNA from L. innocua with a limit of detection of ~2 × 104 cells/mL, which agrees with prior studies. Total time for this assay can be constrained to <2.5 h because a timely culture enrichment period is not necessary. Furthermore, the electrochemical detection assay can be performed with hand-held electronics, allowing this platform to be adopted for near-line monitoring systems.
Keywords: Listeria innocua; Listeria monocytogenes; biosensor; flow-through transducer; foodborne pathogen; graphite felt; rapid detection.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
A molecular beacon-based duplex real-time polymerase chain reaction assay for simultaneous detection of Escherichia coli O157:H7 and Listeria monocytogenes in milk and milk products.Foodborne Pathog Dis. 2009 Dec;6(10):1195-201. doi: 10.1089/fpd.2009.0310. Foodborne Pathog Dis. 2009. PMID: 19735201
-
Detection of Escherichia coli O157:H7 and Listeria monocytogenes in beef products by real-time polymerase chain reaction.Foodborne Pathog Dis. 2004 Winter;1(4):231-40. doi: 10.1089/fpd.2004.1.231. Foodborne Pathog Dis. 2004. PMID: 15992285
-
A duplex qPCR for the simultaneous detection of Escherichia coli O157:H7 and Listeria monocytogenes using LNA probes.Lett Appl Microbiol. 2015 Jul;61(1):20-7. doi: 10.1111/lam.12427. Epub 2015 May 10. Lett Appl Microbiol. 2015. PMID: 25869996
-
Biosensor for the detection of Listeria monocytogenes: emerging trends.Crit Rev Microbiol. 2018 Sep;44(5):590-608. doi: 10.1080/1040841X.2018.1473331. Epub 2018 May 23. Crit Rev Microbiol. 2018. PMID: 29790396 Review.
-
A review of the ecology, genomics, and stress response of Listeria innocua and Listeria monocytogenes.Crit Rev Food Sci Nutr. 2012;52(8):712-25. doi: 10.1080/10408398.2010.507909. Crit Rev Food Sci Nutr. 2012. PMID: 22591342 Review.
Cited by
-
Nanomaterial-Based Fluorescent Biosensor for Food Safety Analysis.Biosensors (Basel). 2022 Nov 23;12(12):1072. doi: 10.3390/bios12121072. Biosensors (Basel). 2022. PMID: 36551039 Free PMC article. Review.
-
Novel Approaches to Environmental Monitoring and Control of Listeria monocytogenes in Food Production Facilities.Foods. 2022 Jun 15;11(12):1760. doi: 10.3390/foods11121760. Foods. 2022. PMID: 35741961 Free PMC article. Review.
-
Beyond antimicrobial resistance: MATE-type efflux pump FepA contributes to flagellum formation and virulence in Listeria monocytogenes.Appl Environ Microbiol. 2025 Jul 23;91(7):e0046225. doi: 10.1128/aem.00462-25. Epub 2025 Jun 10. Appl Environ Microbiol. 2025. PMID: 40492698 Free PMC article.
-
Revolutionizing food safety with electrochemical biosensors for rapid and portable pathogen detection.Braz J Microbiol. 2024 Sep;55(3):2511-2525. doi: 10.1007/s42770-024-01427-6. Epub 2024 Jun 26. Braz J Microbiol. 2024. PMID: 38922532 Free PMC article. Review.
-
Magnetic capture device for large volume sample analysis.PLoS One. 2024 Feb 9;19(2):e0297806. doi: 10.1371/journal.pone.0297806. eCollection 2024. PLoS One. 2024. PMID: 38335195 Free PMC article.
References
-
- Rodrigues C., da Silva A.L.B.R., Dunn L.L. Factors Impacting the Prevalence of Foodborne Pathogens in Agricultural Water Sources in the Southeastern United States. Water. 2020;12:51. doi: 10.3390/w12010051. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

