Effects of Amino Acid Side-Chain Length and Chemical Structure on Anionic Polyglutamic and Polyaspartic Acid Cellulose-Based Polyelectrolyte Brushes
- PMID: 34071693
- PMCID: PMC8199235
- DOI: 10.3390/polym13111789
Effects of Amino Acid Side-Chain Length and Chemical Structure on Anionic Polyglutamic and Polyaspartic Acid Cellulose-Based Polyelectrolyte Brushes
Abstract
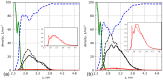
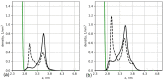
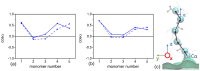
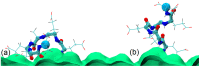
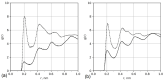
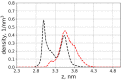
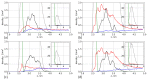
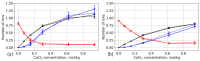
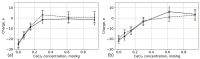
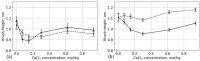
We used atomistic molecular dynamics (MD) simulations to study polyelectrolyte brushes based on anionic α,L-glutamic acid and α,L-aspartic acid grafted on cellulose in the presence of divalent CaCl2 salt at different concentrations. The motivation is to search for ways to control properties such as sorption capacity and the structural response of the brush to multivalent salts. For this detailed understanding of the role of side-chain length, the chemical structure and their interplay are required. It was found that in the case of glutamic acid oligomers, the longer side chains facilitate attractive interactions with the cellulose surface, which forces the grafted chains to lie down on the surface. The additional methylene group in the side chain enables side-chain rotation, enhancing this effect. On the other hand, the shorter and more restricted side chains of aspartic acid oligomers prevent attractive interactions to a large degree and push the grafted chains away from the surface. The difference in side-chain length also leads to differences in other properties of the brush in divalent salt solutions. At a low grafting density, the longer side chains of glutamic acid allow the adsorbed cations to be spatially distributed inside the brush resulting in a charge inversion. With an increase in grafting density, the difference in the total charge of the aspartic and glutamine brushes disappears, but new structural features appear. The longer sides allow for ion bridging between the grafted chains and the cellulose surface without a significant change in main-chain conformation. This leads to the brush structure being less sensitive to changes in salt concentration.
Keywords: cellulose; mineralization; molecular dynamics simulation; poly(amino acids); poly-(α,L-aspartic acid); poly-(α,L-glutamic acid); polyelectrolyte brushes.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Chen W.-L., Cordero R., Tran H., Ober C.K. 50th Anniversary Perspective: Polymer Brushes: Novel Surfaces for Future Materials. Macromolecules. 2017;50:4089–4113. doi: 10.1021/acs.macromol.7b00450. - DOI
-
- Wilts E.M., Herzberger J., Long T.E. Addressing water scarcity: Cationic polyelectrolytes in water treatment and purification. Polym. Int. 2018;67:799–814. doi: 10.1002/pi.5569. - DOI
-
- Migahed M., Rashwan S., Kamel M., Habib R. Synthesized polyaspartic acid derivatives as corrosion and scale inhibitors in desalination operations. Cogent Eng. 2017;4:1366255. doi: 10.1080/23311916.2017.1366255. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

