Acute and Chronic Pain from Facial Skin and Oral Mucosa: Unique Neurobiology and Challenging Treatment
- PMID: 34071720
- PMCID: PMC8198570
- DOI: 10.3390/ijms22115810
Acute and Chronic Pain from Facial Skin and Oral Mucosa: Unique Neurobiology and Challenging Treatment
Abstract
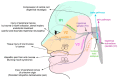
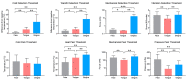
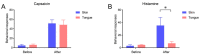
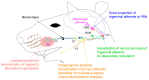
The oral cavity is a portal into the digestive system, which exhibits unique sensory properties. Like facial skin, the oral mucosa needs to be exquisitely sensitive and selective, in order to detect harmful toxins versus edible food. Chemosensation and somatosensation by multiple receptors, including transient receptor potential channels, are well-developed to meet these needs. In contrast to facial skin, however, the oral mucosa rarely exhibits itch responses. Like the gut, the oral cavity performs mechanical and chemical digestion. Therefore, the oral mucosa needs to be insensitive, to some degree, in order to endure noxious irritation. Persistent pain from the oral mucosa is often due to ulcers, involving both tissue injury and infection. Trigeminal nerve injury and trigeminal neuralgia produce intractable pain in the orofacial skin and the oral mucosa, through mechanisms distinct from those seen in the spinal area, which is particularly difficult to predict or treat. The diagnosis and treatment of idiopathic chronic pain, such as atypical odontalgia (idiopathic painful trigeminal neuropathy or post-traumatic trigeminal neuropathy) and burning mouth syndrome, remain especially challenging. The central integration of gustatory inputs might modulate chronic oral and facial pain. A lack of pain in chronic inflammation inside the oral cavity, such as chronic periodontitis, involves the specialized functioning of oral bacteria. A more detailed understanding of the unique neurobiology of pain from the orofacial skin and the oral mucosa should help us develop novel methods for better treating persistent orofacial pain.
Keywords: chronic pain; mucosa pain; orofacial pain.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Bradley R.M. Essentials of Oral Physiology. Mosby; St. Louis, MO, USA: 1995.
-
- Dubner R. The Neural Basis of Oral and Facial Function. Springer; Boston, MA, USA: 1978.
-
- Zhao N.N., Whittle T., Murray G.M., Peck C.C. The effects of capsaicin-induced intraoral mucosal pain on jaw movements in humans. J. Orofac. Pain. 2012;26:277–287. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

