MSC Manufacturing for Academic Clinical Trials: From a Clinical-Grade to a Full GMP-Compliant Process
- PMID: 34073206
- PMCID: PMC8227789
- DOI: 10.3390/cells10061320
MSC Manufacturing for Academic Clinical Trials: From a Clinical-Grade to a Full GMP-Compliant Process
Abstract
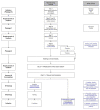
Following European regulation 1394/2007, mesenchymal stromal cell (MSCs) have become an advanced therapy medicinal product (ATMP) that must be produced following the good manufacturing practice (GMP) standards. We describe the upgrade of our existing clinical-grade MSC manufacturing process to obtain GMP certification. Staff organization, premises/equipment qualification and monitoring, raw materials management, starting materials, technical manufacturing processes, quality controls, and the release, thawing and infusion were substantially reorganized. Numerous studies have been carried out to validate cultures and demonstrate the short-term stability of fresh or thawed products, as well their stability during long-term storage. Detailed results of media simulation tests, validation runs and early MSC batches are presented. We also report the validation of a new variant of the process aiming to prepare fresh MSCs for the treatment of specific lesions of Crohn's disease by local injection. In conclusion, we have successfully ensured the adaptation of our clinical-grade MSC production process to the GMP requirements. The GMP manufacturing of MSC products is feasible in the academic setting for a limited number of batches with a significant cost increase, but moving to large-scale production necessary for phase III trials would require the involvement of industrial partners.
Keywords: ATMP manufacturing; GMP; MSC; cell therapy; mesenchymal stromal cells.
Conflict of interest statement
C.L., A.B., V.B., E.B. and Y.B. report no conflict of interest.
Figures
References
-
- Panes J., Garcia-Olmo D., Van A.G., Colombel J.F., Reinisch W., Baumgart D.C., Dignass A., Nachury M., Ferrante M., Kazemi-Shirazi L., et al. Expanded allogeneic adipose-derived mesenchymal stem cells (Cx601) for complex perianal fistulas in Crohn’s disease: A phase 3 randomised, double-blind controlled trial. Lancet. 2016;388:1281–1290. doi: 10.1016/S0140-6736(16)31203-X. - DOI - PubMed
-
- Munneke J.M., Spruit M.J., Cornelissen A.S., Hoeven V.V., Voermans C., Hazenberg M.D. The Potential of Mesenchymal Stromal Cells as Treatment for Severe Steroid-Refractory Acute Graft-Versus-Host Disease: A Critical Review of the Literature. Transplantation. 2016;100:2309–2314. doi: 10.1097/TP.0000000000001029. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

