Short-Term Soil Flushing with Tannic Acid and Its Effect on Metal Mobilization and Selected Properties of Calcareous Soil
- PMID: 34073355
- PMCID: PMC8197821
- DOI: 10.3390/ijerph18115698
Short-Term Soil Flushing with Tannic Acid and Its Effect on Metal Mobilization and Selected Properties of Calcareous Soil
Abstract
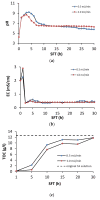
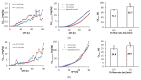
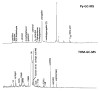
Cadmium, Cu, Ni, Pb, and Zn removal via soil flushing with tannic acid (TA) as a plant biosurfactant was studied. The soil was treated for 30 h in a column reactor at a constant TA concentration and pH (3%, pH 4) and at variable TA flow rates (0.5 mL/min or 1 mL/min). In the soil leachates, pH, electrical conductivity (EC), total dissolved organic carbon, and metal concentrations were monitored. Before and after flushing, soil pH, EC, organic matter content, and cation exchange capacity (CEC) were determined. To analyze the organic matter composition, pyrolysis as well as thermally assisted hydrolysis and methylation coupled with gas chromatography-mass spectrometry were used. Metal fractionation in unflushed and flushed soil was analyzed using a modified sequential extraction method. The data on cumulative metal removal were analyzed using OriginPro 8.0 software (OriginLab Corporation, Northampton, MA, USA) and were fitted to 4-parameter logistic sigmoidal model. It was found that flushing time had a stronger influence on metal removal than flow rate. The overall efficiency of metal removal (expressed as the ratio between flushed metal concentration and total metal concentration in soil) at the higher flow rate decreased in this order: Cd (86%) > Ni (44%) > Cu (29%) ≈ Zn (26%) > Pb (15%). Metals were removed from the exchangeable fraction and redistributed into the reducible fraction. After flushing, the soil had a lower pH, EC, and CEC; a higher organic matter content; the composition of the organic matter had changed (incorporation of TA structures). Our results prove that soil flushing with TA is a promising approach to decrease metal concentration in soil and to facilitate carbon sequestration in soil.
Keywords: heavy metals; soil column; tannic acid sorption.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Correlation of the partitioning of dissolved organic matter fractions with the desorption of Cd, Cu, Ni, Pb and Zn from 18 Dutch soils.Environ Int. 2002 Nov;28(5):401-10. doi: 10.1016/s0160-4120(02)00065-x. Environ Int. 2002. PMID: 12437290
-
[Speciation and bioavailability of heavy metals in paddy soil irrigated by acid mine drainage].Huan Jing Ke Xue. 2009 Mar 15;30(3):900-6. Huan Jing Ke Xue. 2009. PMID: 19432348 Chinese.
-
Effect of bamboo and rice straw biochars on the mobility and redistribution of heavy metals (Cd, Cu, Pb and Zn) in contaminated soil.J Environ Manage. 2017 Jan 15;186(Pt 2):285-292. doi: 10.1016/j.jenvman.2016.05.068. Epub 2016 Jun 2. J Environ Manage. 2017. PMID: 27264699
-
Chelant extraction of heavy metals from contaminated soils.J Hazard Mater. 1999 Apr 23;66(1-2):151-210. doi: 10.1016/s0304-3894(99)00010-2. J Hazard Mater. 1999. PMID: 10379036 Review.
-
A critical review of the bioavailability and impacts of heavy metals in municipal solid waste composts compared to sewage sludge.Environ Int. 2009 Jan;35(1):142-56. doi: 10.1016/j.envint.2008.06.009. Epub 2008 Aug 8. Environ Int. 2009. PMID: 18691760 Review.
Cited by
-
The Application of Natural Phenolic Substances as Antimicrobial Agents in Agriculture and Food Industry.Foods. 2025 May 26;14(11):1893. doi: 10.3390/foods14111893. Foods. 2025. PMID: 40509420 Free PMC article. Review.
-
Remediation of Smelter Contaminated Soil by Sequential Washing Using Biosurfactants.Int J Environ Res Public Health. 2021 Dec 7;18(24):12875. doi: 10.3390/ijerph182412875. Int J Environ Res Public Health. 2021. PMID: 34948484 Free PMC article.
References
-
- Khalid S., Shahid M., Niazi N.K., Murtaza B., Bibi I., Dumat C. A comparison of technologies for remediation of heavy HM contaminated soils. J. Geochem. Explor. 2017;182:247–268. doi: 10.1016/j.gexplo.2016.11.021. - DOI
-
- Alloway B.J. Heavy HMs in Soils. Blackie Academic and Professional, Chapman and Hall; London, UK: 1995. 368p
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

