Propellanes as Rigid Scaffolds for the Stereodefined Attachment of σ-Pharmacophoric Structural Elements to Achieve σ Affinity
- PMID: 34073622
- PMCID: PMC8199339
- DOI: 10.3390/ijms22115685
Propellanes as Rigid Scaffolds for the Stereodefined Attachment of σ-Pharmacophoric Structural Elements to Achieve σ Affinity
Abstract
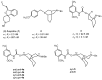
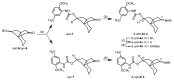
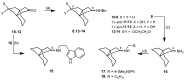
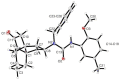
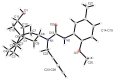
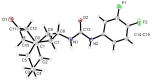
Following the concept of conformationally restriction of ligands to achieve high receptor affinity, we exploited the propellane system as rigid scaffold allowing the stereodefined attachment of various substituents. Three types of ligands were designed, synthesized and pharmacologically evaluated as σ1 receptor ligands. Propellanes with (1) a 2-methoxy-5-methylphenylcarbamate group at the "left" five-membered ring and various amino groups on the "right" side; (2) benzylamino or analogous amino moieties on the "right" side and various substituents at the left five-membered ring and (3) various urea derivatives at one five-membered ring were investigated. The benzylamino substituted carbamate syn,syn-4a showed the highest σ1 affinity within the group of four stereoisomers emphasizing the importance of the stereochemistry. The cyclohexylmethylamine 18 without further substituents at the propellane scaffold revealed unexpectedly high σ1 affinity (Ki = 34 nM) confirming the relevance of the bioisosteric replacement of the benzylamino moiety by the cyclohexylmethylamino moiety. Reduction of the distance between the basic amino moiety and the "left" hydrophobic region by incorporation of the amino moiety into the propellane scaffold resulted in azapropellanes with particular high σ1 affinity. As shown for the propellanamine 18, removal of the carbamate moiety increased the σ1 affinity of 9a (Ki = 17 nM) considerably. Replacement of the basic amino moiety by H-bond forming urea did not lead to potent σ ligands. According to molecular dynamics simulations, both azapropellanes anti-5 and 9a as well as propellane 18 adopt binding poses at the σ1 receptor, which result in energetic values correlating well with their different σ1 affinities. The affinity of the ligands is enthalpy driven. The additional interactions of the carbamate moiety of anti-5 with the σ1 receptor protein cannot compensate the suboptimal orientations of the rigid propellane and its N-benzyl moiety within the σ1 receptor-binding pocket, which explains the higher σ1 affinity of the unsubstituted azapropellane 9a.
Keywords: X-ray crystal structures; azapropellanes; molecular dynamics; molecular interactions; propellanes; rigidity; selectivity; stereochemistry; σ receptors.
Conflict of interest statement
The authors have no conflict of interests to declare.
Figures






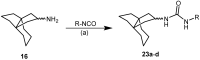
Similar articles
-
Synthesis of 3-aza[4.4.3]propellanes with high σ1 receptor affinity.Bioorg Med Chem. 2018 May 1;26(8):1705-1712. doi: 10.1016/j.bmc.2018.02.019. Epub 2018 Feb 15. Bioorg Med Chem. 2018. PMID: 29482951
-
Synthesis and Structure-Affinity Relationships of Spirocyclic Benzopyrans with Exocyclic Amino Moiety.J Med Chem. 2019 Apr 25;62(8):4204-4217. doi: 10.1021/acs.jmedchem.9b00449. Epub 2019 Apr 12. J Med Chem. 2019. PMID: 30939014
-
Chemoenzymatic synthesis of 2,6-disubstituted tetrahydropyrans with high σ1 receptor affinity, antitumor and analgesic activity.Eur J Med Chem. 2021 Jul 5;219:113443. doi: 10.1016/j.ejmech.2021.113443. Epub 2021 Apr 20. Eur J Med Chem. 2021. PMID: 33901806
-
Pharmacophore models and development of spirocyclic ligands for σ1 receptors.Curr Pharm Des. 2012;18(7):930-7. doi: 10.2174/138161212799436548. Curr Pharm Des. 2012. PMID: 22288413 Review.
-
[18F]N-(2-benzofuranylmethyl)-N'-[4-(2-fluoroethoxy)benzyl]piperazine.2012 May 10 [updated 2012 Jun 27]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. 2012 May 10 [updated 2012 Jun 27]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. PMID: 22764386 Free Books & Documents. Review.
References
-
- Martin W.R., Eades C.G., Thompson J.A., Huppler R.E., Gilbert P.E. The effects of morphine- and nalorphine- like drugs in the nondependent and morphine-dependent chronic spinal dog. J. Pharmacol. Exp. Ther. 1976;197:517–532. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

