Neurotrophins as Key Regulators of Cell Metabolism: Implications for Cholesterol Homeostasis
- PMID: 34073639
- PMCID: PMC8198482
- DOI: 10.3390/ijms22115692
Neurotrophins as Key Regulators of Cell Metabolism: Implications for Cholesterol Homeostasis
Abstract
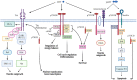
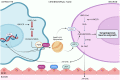
Neurotrophins constitute a family of growth factors initially characterized as predominant mediators of nervous system development, neuronal survival, regeneration and plasticity. Their biological activity is promoted by the binding of two different types of receptors, leading to the generation of multiple and variegated signaling cascades in the target cells. Increasing evidence indicates that neurotrophins are also emerging as crucial regulators of metabolic processes in both neuronal and non-neuronal cells. In this context, it has been reported that neurotrophins affect redox balance, autophagy, glucose homeostasis and energy expenditure. Additionally, the trophic support provided by these secreted factors may involve the regulation of cholesterol metabolism. In this review, we examine the neurotrophins' signaling pathways and their effects on metabolism by critically discussing the most up-to-date information. In particular, we gather experimental evidence demonstrating the impact of these growth factors on cholesterol metabolism.
Keywords: BDNF; NGF; TrkA; TrkB; cholesterol; metabolism; neurotrophins; p75NTR.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Differential dependency of cutaneous mechanoreceptors on neurotrophins, trk receptors, and P75 LNGFR.Dev Biol. 1997 Oct 1;190(1):94-116. doi: 10.1006/dbio.1997.8658. Dev Biol. 1997. PMID: 9331334
-
Type I interferons up-regulate the expression and signalling of p75 NTR/TrkA receptor complex in differentiated human SH-SY5Y neuroblastoma cells.Neuropharmacology. 2014 Apr;79:321-34. doi: 10.1016/j.neuropharm.2013.12.002. Epub 2013 Dec 11. Neuropharmacology. 2014. PMID: 24333329
-
Neurotrophins and Neuropathic Pain: Role in Pathobiology.Molecules. 2015 Jun 9;20(6):10657-88. doi: 10.3390/molecules200610657. Molecules. 2015. PMID: 26065639 Free PMC article. Review.
-
Neurotrophins' Modulation by Olive Polyphenols.Curr Med Chem. 2016;23(28):3189-3197. doi: 10.2174/0929867323666160627104022. Curr Med Chem. 2016. PMID: 27356540 Review.
-
Functional switch from pro-neurotrophins to mature neurotrophins.Curr Protein Pept Sci. 2013 Nov;14(7):617-25. doi: 10.2174/1389203711209070658. Curr Protein Pept Sci. 2013. PMID: 24106968 Review.
Cited by
-
The role of DHCR24 in the pathogenesis of AD: re-cognition of the relationship between cholesterol and AD pathogenesis.Acta Neuropathol Commun. 2022 Mar 16;10(1):35. doi: 10.1186/s40478-022-01338-3. Acta Neuropathol Commun. 2022. PMID: 35296367 Free PMC article. Review.
-
Brain-Derived Neurotrophic Factor - The Protective Agent Against Neurological Disorders.CNS Neurol Disord Drug Targets. 2024;23(3):353-366. doi: 10.2174/1871527322666230607110617. CNS Neurol Disord Drug Targets. 2024. PMID: 37287291 Free PMC article.
-
Leptin Receptor (rs1137101) and Brain-Derived Neurotrophic Factor (rs925946) Gene Variants Are Associated with Obesity in the Early- but Not in the Late-Onset Population of Hungarian Psoriatic Patients.Life (Basel). 2021 Oct 14;11(10):1086. doi: 10.3390/life11101086. Life (Basel). 2021. PMID: 34685457 Free PMC article.
-
Candidate Key Proteins in Tinnitus: A Bioinformatic Study of Synaptic Transmission in Spiral Ganglion Neurons.Cell Mol Neurobiol. 2023 Nov;43(8):4189-4207. doi: 10.1007/s10571-023-01405-w. Epub 2023 Sep 22. Cell Mol Neurobiol. 2023. PMID: 37736859 Free PMC article.
-
The Effect of Endurance Training on Serum BDNF Levels in the Chronic Post-Stroke Phase: Current Evidence and Qualitative Systematic Review.J Clin Med. 2022 Jun 20;11(12):3556. doi: 10.3390/jcm11123556. J Clin Med. 2022. PMID: 35743624 Free PMC article. Review.
References
-
- Chaldakov G.N., Tonchev A.B., Aloe L. NGF and BDNF: From nerves to adipose tissue, from neurokines to metabokines. Riv. Psichiatr. 2010;44:79–87. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

