Detection of Cryptic Fragile X Full Mutation Alleles by Southern Blot in a Female and Her Foetal DNA via Chorionic Villus Sampling, Complicated by Mosaicism for 45,X0/46,XX/47,XXX
- PMID: 34073864
- PMCID: PMC8225079
- DOI: 10.3390/genes12060798
Detection of Cryptic Fragile X Full Mutation Alleles by Southern Blot in a Female and Her Foetal DNA via Chorionic Villus Sampling, Complicated by Mosaicism for 45,X0/46,XX/47,XXX
Abstract
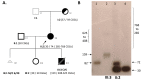
We describe a female with a 72 CGG FMR1 premutation (PM) (CGG 55-199) and family history of fragile X syndrome (FXS), referred for prenatal testing. The proband had a high risk of having an affected pregnancy with a full mutation allele (FM) (CGG > 200), that causes FXS through hypermethylation of the FMR1 promoter. The CGG sizing analysis in this study used AmplideX triplet repeat primed polymerase chain reaction (TP-PCR) and long-range methylation sensitive PCR (mPCR). These methods detected a 73 CGG PM allele in the proband's blood, and a 164 CGG PM allele in her male cultured chorionic villus sample (CVS). In contrast, the Southern blot analysis showed mosaicism for: (i) a PM (71 CGG) and an FM (285-768 CGG) in the proband's blood, and (ii) a PM (165 CGG) and an FM (408-625 CGG) in the male CVS. The FMR1 methylation analysis, using an EpiTYPER system in the proband, showed levels in the range observed for mosaic Turner syndrome. This was confirmed by molecular and cytogenetic karyotyping, identifying 45,X0/46,XX/47,XXX lines. In conclusion, this case highlights the importance of Southern blot in pre- and postnatal testing for presence of an FM, which was not detected using AmplideX TP-PCR or mPCR in the proband and her CVS.
Keywords: 45,X0/46,XX/47,XXX; CGG; FMR1; X syndrome; cultured chorionic villus; fragile; full mutation; mosaicism; sample; southern blot.
Conflict of interest statement
David E. Godler is named as an inventor on patent applications (PCT/AU2010/000169 and PCT/AU2014/00004) related to the technology described in this article. The other authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
Qualitative assessment of FMR1 (CGG)n triplet repeat status in normal, intermediate, premutation, full mutation, and mosaic carriers in both sexes: implications for fragile X syndrome carrier and newborn screening.Genet Med. 2010 Mar;12(3):162-73. doi: 10.1097/GIM.0b013e3181d0d40e. Genet Med. 2010. PMID: 20168238
-
Clinical and Molecular Differences between 4-Year-Old Monozygous Male Twins Mosaic for Normal, Premutation and Fragile X Full Mutation Alleles.Genes (Basel). 2019 Apr 5;10(4):279. doi: 10.3390/genes10040279. Genes (Basel). 2019. PMID: 30959842 Free PMC article.
-
Abnormally Methylated FMR1 in Absence of a Detectable Full Mutation in a U.S.A Patient Cohort Referred for Fragile X Testing.Sci Rep. 2019 Oct 25;9(1):15315. doi: 10.1038/s41598-019-51618-7. Sci Rep. 2019. PMID: 31653898 Free PMC article.
-
Somatic Instability Leading to Mosaicism in Fragile X Syndrome and Associated Disorders: Complex Mechanisms, Diagnostics, and Clinical Relevance.Int J Mol Sci. 2024 Dec 21;25(24):13681. doi: 10.3390/ijms252413681. Int J Mol Sci. 2024. PMID: 39769443 Free PMC article. Review.
-
Clinical and molecular characteristics of FMR1 microdeletion in patient with fragile X syndrome and review of the literature.Clin Chim Acta. 2024 Jan 15;553:117728. doi: 10.1016/j.cca.2023.117728. Epub 2023 Dec 23. Clin Chim Acta. 2024. PMID: 38142803 Review.
Cited by
-
C9orf72 repeat expansion creates the unstable folate-sensitive fragile site FRA9A.NAR Mol Med. 2024 Nov 12;1(4):ugae019. doi: 10.1093/narmme/ugae019. eCollection 2024 Oct. NAR Mol Med. 2024. PMID: 39669124 Free PMC article.
-
Fragile sites, chromosomal lesions, tandem repeats, and disease.Front Genet. 2022 Nov 17;13:985975. doi: 10.3389/fgene.2022.985975. eCollection 2022. Front Genet. 2022. PMID: 36468036 Free PMC article. Review.
-
Editorial for the Fragile X Syndrome Genetics Special Issue: May 2023.Genes (Basel). 2023 May 25;14(6):1148. doi: 10.3390/genes14061148. Genes (Basel). 2023. PMID: 37372328 Free PMC article.
References
-
- Godler D.E., Inaba Y., Schwartz C.E., Bui Q.M., Shi E.Z., Li X., Herlihy A.S., Skinner C., Hagerman R.J., Francis D., et al. Detection of skewed X-chromosome inactivation in Fragile X syndrome and X chromosome aneuploidy using quantitative melt analysis. Expert Rev. Mol. Med. 2015;17:e13. doi: 10.1017/erm.2015.11. - DOI - PMC - PubMed
-
- Godler D.E., Tassone F., Loesch D.Z., Taylor A.K., Gehling F., Hagerman R.J., Burgess T., Ganesamoorthy D., Hennerich D., Gordon L., et al. Methylation of novel markers of fragile X alleles is inversely correlated with FMRP expression and FMR1 activation ratio. Hum. Mol. Genet. 2010;19:1618–1632. doi: 10.1093/hmg/ddq037. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

