Assessing the Interactions of Statins with Human Adenylate Kinase Isoenzyme 1: Fluorescence and Enzyme Kinetic Studies
- PMID: 34073952
- PMCID: PMC8197361
- DOI: 10.3390/ijms22115541
Assessing the Interactions of Statins with Human Adenylate Kinase Isoenzyme 1: Fluorescence and Enzyme Kinetic Studies
Abstract
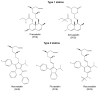
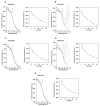
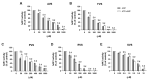
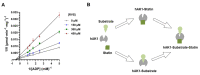
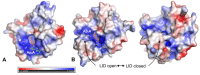
Statins are the most effective cholesterol-lowering drugs. They also exert many pleiotropic effects, including anti-cancer and cardio- and neuro-protective. Numerous nano-sized drug delivery systems were developed to enhance the therapeutic potential of statins. Studies on possible interactions between statins and human proteins could provide a deeper insight into the pleiotropic and adverse effects of these drugs. Adenylate kinase (AK) was found to regulate HDL endocytosis, cellular metabolism, cardiovascular function and neurodegeneration. In this work, we investigated interactions between human adenylate kinase isoenzyme 1 (hAK1) and atorvastatin (AVS), fluvastatin (FVS), pravastatin (PVS), rosuvastatin (RVS) and simvastatin (SVS) with fluorescence spectroscopy. The tested statins quenched the intrinsic fluorescence of hAK1 by creating stable hAK1-statin complexes with the binding constants of the order of 104 M-1. The enzyme kinetic studies revealed that statins inhibited hAK1 with significantly different efficiencies, in a noncompetitive manner. Simvastatin inhibited hAK1 with the highest yield comparable to that reported for diadenosine pentaphosphate, the only known hAK1 inhibitor. The determined AK sensitivity to statins differed markedly between short and long type AKs, suggesting an essential role of the LID domain in the AK inhibition. Our studies might open new horizons for the development of new modulators of short type AKs.
Keywords: adenylate kinase; atorvastatin; fluorescence spectroscopy; fluvastatin; inhibitors; pleiotropic effect; pravastatin; rosuvastatin; simvastatin.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Characterization of interactions of simvastatin, pravastatin, fluvastatin, and pitavastatin with bovine serum albumin: multiple spectroscopic and molecular docking.J Biomol Struct Dyn. 2017 May;35(7):1529-1546. doi: 10.1080/07391102.2016.1188416. Epub 2016 Aug 3. J Biomol Struct Dyn. 2017. PMID: 27484332
-
Low-density lipoprotein cholesterol (LDL-C) levels and LDL-C goal attainment among elderly patients treated with rosuvastatin compared with other statins in routine clinical practice.Am J Geriatr Pharmacother. 2007 Sep;5(3):185-94. doi: 10.1016/j.amjopharm.2007.10.002. Am J Geriatr Pharmacother. 2007. PMID: 17996658
-
Comparison of the Effects of Statins on A549 Nonsmall-Cell Lung Cancer Cell Line Lipids Using Fourier Transform Infrared Spectroscopy: Rosuvastatin Stands Out.Lipids. 2021 May;56(3):289-299. doi: 10.1002/lipd.12296. Epub 2021 Feb 21. Lipids. 2021. PMID: 33611813
-
Pharmacodynamics and pharmacokinetics of the HMG-CoA reductase inhibitors. Similarities and differences.Clin Pharmacokinet. 1997 May;32(5):403-25. doi: 10.2165/00003088-199732050-00005. Clin Pharmacokinet. 1997. PMID: 9160173 Review.
-
New insights into the pharmacodynamic and pharmacokinetic properties of statins.Pharmacol Ther. 1999 Dec;84(3):413-28. doi: 10.1016/s0163-7258(99)00045-5. Pharmacol Ther. 1999. PMID: 10665838 Review.
Cited by
-
Identification of Serum Biomarkers for Early-Stage Knee Osteoarthritis Using Proteomics in a Murine Model of Osteoarthritis.Cartilage. 2025 Aug 8:19476035251363443. doi: 10.1177/19476035251363443. Online ahead of print. Cartilage. 2025. PMID: 40781794 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

