Adjunctive Perampanel in Older Patients With Epilepsy: A Multicenter Study of Clinical Practice
- PMID: 34075567
- PMCID: PMC8266697
- DOI: 10.1007/s40266-021-00865-3
Adjunctive Perampanel in Older Patients With Epilepsy: A Multicenter Study of Clinical Practice
Abstract
Background: Clinical data regarding use of newer antiseizure medications (ASMs) in an older population are limited. In randomized-controlled, placebo-controlled trials, older patients are under-represented, and protocols deviate markedly from routine clinical practice, limiting the external validity of results. Studies performed in a naturalistic setting are a useful complement to characterize the drug profile. Perampanel is a third-generation ASM and the first and only non-competitive alfa-amino-3-hydroxyl-5-methyl-4-isoxazole-propionate receptor antagonist.
Objective: The aim of this study was to assess the effectiveness and tolerability of adjunctive perampanel over a 1-year period in a population of older patients with epilepsy treated in a real-world setting.
Methods: Older (≥ 65 years of age) patients prescribed add-on perampanel at 12 Italian epilepsy centers were retrospectively identified. Seizure occurrence, adverse events (AEs), and drug withdrawal were analyzed. Effectiveness outcomes included the rates of seizure response (≥ 50% reduction in baseline monthly seizure frequency), seizure freedom, and treatment discontinuation. Safety and tolerability outcomes were the rate of treatment discontinuation due to AEs and the incidence of AEs.
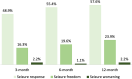
Results: A total of 92 patients with a median age of 69 (range 65-88) years were included. The median daily dose of perampanel at 12 months was 6 mg (interquartile range 4-6 mg). At 12 months, 53 (57.6%) patients were seizure responders, and 22 (23.9%) patients were seizure free. Twenty (21.7%) patients discontinued perampanel; the reasons for treatment withdrawal were insufficient efficacy (n = 6/20; 30.0%), AEs (n = 12/20; 60.0%), and a combination of both (n = 2/20; 10%). The most common AEs included irritability (8.7%), somnolence (4.3%), and dizziness/vertigo (4.3%). The rate of behavioral and psychiatric AEs was higher in patients with history of psychiatric comorbidities (p = 0.044). There were no differences in the occurrence of behavioral and psychiatric AEs according to the concomitant use of levetiracetam (p = 0.776) and history of cognitive decline (p = 0.332).
Conclusions: Adjunctive perampanel was associated with improvement in seizure control and good tolerability in a real-life setting and can represent a viable therapeutic option in older patients with epilepsy.
Conflict of interest statement
Simona Lattanzi has received speaker's or consultancy fees from Eisai, GW Pharmaceuticals, and UCB Pharma and has served on advisory boards for Angelini, Arvelle Therapeutics, BIAL, and GW Pharmaceuticals. Valentina Chiesa has received speaker's or consultancy fees from Eisai, UCB Pharma, and Livanova. Fedele Dono has received speaker's fees from Eisai. Maria Paola Canevini has received speaker’s or consultancy fees from Bial, EISAI, Italfarmaco, Sanofi, and UCB. All other authors have nothing to disclose.
Figures
References
-
- Rohracher A, Kalss G, Kuchukhidze G, Neuray C, Leitinger M, Höfler J, Kreidenhuber R, Rossini F, Volna K, Mauritz M, Poppert N, Lattanzi S, Brigo F, Trinka E. New anti-seizure medication for elderly epilepsy patients—a critical narrative review. Expert Opin Pharmacother. 2021;22:621–634. doi: 10.1080/14656566.2020.1843636. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

