Bivalent Conjugate Vaccine Induces Dual Immunogenic Response That Attenuates Heroin and Fentanyl Effects in Mice
- PMID: 34076427
- PMCID: PMC8603354
- DOI: 10.1021/acs.bioconjchem.1c00179
Bivalent Conjugate Vaccine Induces Dual Immunogenic Response That Attenuates Heroin and Fentanyl Effects in Mice
Abstract
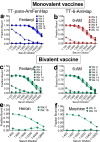
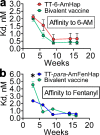
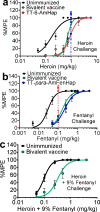
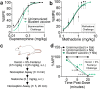
Opioid use disorders and fatal overdose due to consumption of fentanyl-laced heroin remain a major public health menace in the United States. Vaccination may serve as a promising potential remedy to combat accidental overdose and to mitigate the abuse potential of opioids. We previously reported the heroin and fentanyl monovalent vaccines carrying, respectively, a heroin hapten, 6-AmHap, and a fentanyl hapten, para-AmFenHap, conjugated to tetanus toxoid (TT). Herein, we describe the mixing of these antigens to formulate a bivalent vaccine adjuvanted with liposomes containing monophosphoryl lipid A (MPLA) adsorbed on aluminum hydroxide. Immunization of mice with the bivalent vaccine resulted in IgG titers of >105 against both haptens. The polyclonal sera bound heroin, 6-acetylmorphine, morphine, and fentanyl with dissociation constants (Kd) of 0.25 to 0.50 nM. Mice were protected from the anti-nociceptive effects of heroin, fentanyl, and heroin +9% (w/w) fentanyl. No cross-reactivity to methadone and buprenorphine was observed in vivo. Naloxone remained efficacious in immunized mice. These results highlighted the potential of combining TT-6-AmHap and TT-para-AmFenHap to yield an efficacious bivalent vaccine that could ablate heroin and fentanyl effects. This vaccine warrants further testing to establish its potential translatability to humans.
Conflict of interest statement
The authors declare the following competing financial interest(s): GRM, OBT, AS, KCR, AEJ, and EB are inventors of a provision patent application filed by the Henry M. Jackson Foundation for the Advancement of Military Medicine (provisional patent Serial No.: 62/960,187; January 13, 2020).
Figures


Similar articles
-
Efficacy, but not antibody titer or affinity, of a heroin hapten conjugate vaccine correlates with increasing hapten densities on tetanus toxoid, but not on CRM197 carriers.Bioconjug Chem. 2015 Jun 17;26(6):1041-53. doi: 10.1021/acs.bioconjchem.5b00085. Epub 2015 Jun 5. Bioconjug Chem. 2015. PMID: 25970207 Free PMC article.
-
Liposomes containing monophosphoryl lipid A: a potent adjuvant system for inducing antibodies to heroin hapten analogs.Vaccine. 2013 Jun 10;31(26):2804-10. doi: 10.1016/j.vaccine.2013.04.027. Epub 2013 Apr 23. Vaccine. 2013. PMID: 23624097 Free PMC article.
-
Novel Vaccine That Blunts Fentanyl Effects and Sequesters Ultrapotent Fentanyl Analogues.Mol Pharm. 2020 Sep 8;17(9):3447-3460. doi: 10.1021/acs.molpharmaceut.0c00497. Epub 2020 Aug 13. Mol Pharm. 2020. PMID: 32787282 Free PMC article.
-
Adjuvants for vaccines to drugs of abuse and addiction.Vaccine. 2014 Sep 22;32(42):5382-9. doi: 10.1016/j.vaccine.2014.07.085. Epub 2014 Aug 8. Vaccine. 2014. PMID: 25111169 Free PMC article. Review.
-
Conjugate Vaccine Immunotherapy for Substance Use Disorder.Pharmacol Rev. 2017 Jul;69(3):298-315. doi: 10.1124/pr.117.013904. Pharmacol Rev. 2017. PMID: 28634286 Free PMC article. Review.
Cited by
-
Assessment of two brands of fentanyl test strips with 251 synthetic opioids reveals "blind spots" in detection capabilities.Harm Reduct J. 2023 Dec 6;20(1):175. doi: 10.1186/s12954-023-00911-w. Harm Reduct J. 2023. PMID: 38057832 Free PMC article.
-
Preclinical Efficacy and Selectivity of Vaccines Targeting Fentanyl, Alfentanil, Sufentanil, and Acetylfentanyl in Rats.ACS Omega. 2022 May 4;7(19):16584-16592. doi: 10.1021/acsomega.2c00820. eCollection 2022 May 17. ACS Omega. 2022. PMID: 35601290 Free PMC article.
-
The development of opioid vaccines as a novel strategy for the treatment of opioid use disorder and overdose prevention.Int J Neuropsychopharmacol. 2025 Feb 4;28(2):pyaf005. doi: 10.1093/ijnp/pyaf005. Int J Neuropsychopharmacol. 2025. PMID: 39831679 Free PMC article. Review.
-
Conjugation of Multiple Proteins Onto the Surface of PLGA/Lipid Hybrid Nanoparticles.J Biomed Mater Res A. 2025 Jan;113(1):e37807. doi: 10.1002/jbm.a.37807. Epub 2024 Oct 17. J Biomed Mater Res A. 2025. PMID: 39420678
-
Unique Pharmacology, Brain Dysfunction, and Therapeutic Advancements for Fentanyl Misuse and Abuse.Neurosci Bull. 2022 Nov;38(11):1365-1382. doi: 10.1007/s12264-022-00872-3. Epub 2022 May 15. Neurosci Bull. 2022. PMID: 35570233 Free PMC article. Review.
References
-
- National Institute on Drug Abuse (2020). Overdose Death Rates. https://www.drugabuse.gov/related-topics/trends-statistics/overdose-deat... (accessed May 14, 2020).
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

