Arene-Perfluoroarene Interactions in Solution
- PMID: 34077211
- PMCID: PMC8327707
- DOI: 10.1021/acs.joc.1c00921
Arene-Perfluoroarene Interactions in Solution
Abstract
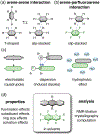
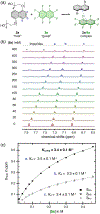
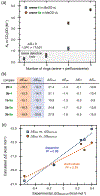
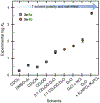
A systematic study of arene-perfluoroarene interactions in solution is presented. Using a combination of NMR titration experiments, X-ray crystallography, and computational analysis, we analyze the effects of fluorination, substituents, ring size, and solvation on the arene-perfluoroarene interaction. We find that fluorination, extension of the π systems, and enhancement of solvent polarity greatly stabilize the stacking energy up to 3 orders of magnitude (Ka = <1 to 6000 M-1), with the highest Ka achieved for the interaction of water-soluble variants of perfluoronaphthalene and anthracene in buffered D2O (pD = 12). Combining computational and experimental results, we conclude that this impressive binding energy is a result of enthalpically favorable electrostatic and dispersion interactions as well as the entropically driven hydrophobic effect. The enhanced understanding of arene-perfluoroarene interactions in aqueous solution sets the stage for the implementation of this abiotic intermolecular interaction in biology and medicine.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




Similar articles
-
X-ray diffraction and theoretical studies for the quantitative assessment of intermolecular arene-perfluoroarene stacking interactions.Chemistry. 2006 Apr 24;12(13):3538-46. doi: 10.1002/chem.200501248. Chemistry. 2006. PMID: 16506260
-
Pressure-Induced Polymerization of Polycyclic Arene-Perfluoroarene Cocrystals: Single Crystal X-ray Diffraction Studies, Reaction Kinetics, and Design of Columnar Hydrofluorocarbons.J Am Chem Soc. 2020 Nov 4;142(44):18907-18923. doi: 10.1021/jacs.0c09021. Epub 2020 Oct 23. J Am Chem Soc. 2020. PMID: 33095990
-
Organometallic ruthenium(II) diamine anticancer complexes: arene-nucleobase stacking and stereospecific hydrogen-bonding in guanine adducts.J Am Chem Soc. 2002 Mar 27;124(12):3064-82. doi: 10.1021/ja017482e. J Am Chem Soc. 2002. PMID: 11902898
-
Intermolecular Interactions between Drugs and Aminoalkyl Methacrylate Copolymer in Solution to Enhance the Concentration of Poorly Water-Soluble Drugs.Chem Pharm Bull (Tokyo). 2019;67(9):906-914. doi: 10.1248/cpb.c18-00849. Chem Pharm Bull (Tokyo). 2019. PMID: 31474728 Review.
-
Orotidine 5'-Monophosphate Decarboxylase: Probing the Limits of the Possible for Enzyme Catalysis.Acc Chem Res. 2018 Apr 17;51(4):960-969. doi: 10.1021/acs.accounts.8b00059. Epub 2018 Mar 29. Acc Chem Res. 2018. PMID: 29595949 Free PMC article. Review.
Cited by
-
Lewis Adduct-Induced Phase Transitions in Polymer/Solvent Mixtures.ACS Polym Au. 2021 Nov 17;2(1):35-41. doi: 10.1021/acspolymersau.1c00024. eCollection 2022 Feb 9. ACS Polym Au. 2021. PMID: 36855742 Free PMC article.
-
Engineering perfluoroarenes for enhanced molecular barrier effect and chirality transfer in solutions.Chem Sci. 2025 Jan 10;16(8):3498-3508. doi: 10.1039/d4sc07859d. eCollection 2025 Feb 19. Chem Sci. 2025. PMID: 39845871 Free PMC article.
-
Enhancement of the Chiroptical Properties of o-OPE through Arene-Perfluoroarene Interactions.Org Lett. 2025 Aug 8;27(31):8459-8463. doi: 10.1021/acs.orglett.5c02277. Epub 2025 Jul 30. Org Lett. 2025. PMID: 40739103 Free PMC article.
-
Selective chiral dimerization and folding driven by arene-perfluoroarene force.Chem Sci. 2023 Nov 29;15(2):618-628. doi: 10.1039/d3sc05212e. eCollection 2024 Jan 3. Chem Sci. 2023. PMID: 38179513 Free PMC article.
-
Tailored Tolane-Perfluorotolane Assembly as Supramolecular Base Pair Replacement in DNA.Angew Chem Int Ed Engl. 2023 Jan 2;62(1):e202214456. doi: 10.1002/anie.202214456. Epub 2022 Dec 1. Angew Chem Int Ed Engl. 2023. PMID: 36344446 Free PMC article.
References
-
- Riley KE; Hobza P Noncovalent Interactions in Biochemistry. Wiley Interdiscip. Rev.: Comput. Mol. Sci 2011, 1 (1), 3–17.
-
- Hobza P;Šponer J Structure, Energetics, and Dynamics of the Nucleic Acid Base Pairs: Nonempirical Ab Initio Calculations. Chem. Rev 1999, 99 (11), 3247–3276. - PubMed
-
- Jurečka P; Hobza P True Stabilization Energies for the Optimal Planar Hydrogen-Bonded and Stacked Structures of Guanine···Cytosine, Adenine···Thymine, and Their 9- and 1-Methyl Derivatives: Complete Basis Set Calculations at the MP2 and CCSD(T) Levels and Comparison with Experiment. J. Am. Chem. Soc 2003, 125 (50), 15608–15613. - PubMed
-
- Salonen LM; Ellermann M; Diederich F Aromatic Rings in Chemical and Biological Recognition: Energetics and Structures. Angew. Chem., Int. Ed 2011, 50 (21), 4808–4842. - PubMed
-
- Müller-Dethlefs K; Hobza P Noncovalent Interactions: A Challenge for Experiment and Theory. Chem. Rev 2000, 100 (1), 143–167. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

