Clinical Value of Consensus Molecular Subtypes in Colorectal Cancer: A Systematic Review and Meta-Analysis
- PMID: 34077519
- PMCID: PMC9002278
- DOI: 10.1093/jnci/djab106
Clinical Value of Consensus Molecular Subtypes in Colorectal Cancer: A Systematic Review and Meta-Analysis
Abstract
Background: The consensus molecular subtypes (CMSs) of colorectal cancer (CRC) capture tumor heterogeneity at the gene-expression level. Currently, a restricted number of molecular features are used to guide treatment for CRC. We summarize the evidence on the clinical value of the CMSs.
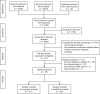
Methods: We systematically identified studies in Medline and Embase that evaluated the prognostic and predictive value of CMSs in CRC patients. A random-effect meta-analysis was performed on prognostic data. Predictive data were summarized.
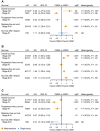
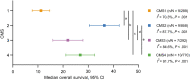
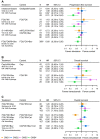
Results: In local disease, CMS4 tumors were associated with worse overall survival (OS) compared with CMS1 (hazard ratio [HR] = 3.28, 95% confidence interval = 1.27 to 8.47) and CMS2 cancers (HR = 2.60, 95% confidence interval = 1.93 to 3.50). In metastatic disease, CMS1 consistently had worse survival than CMS2-4 (OS HR range = 0.33-0.55; progression-free survival HR range = 0.53-0.89). Adjuvant chemotherapy in stage II and III CRC was most beneficial for OS in CMS2 and CMS3 (HR range = 0.16-0.45) and not effective in CMS4 tumors. In metastatic CMS4 cancers, an irinotecan-based regimen improved outcome compared with oxaliplatin (HR range = 0.31-0.72). The addition of bevacizumab seemed beneficial in CMS1, and anti-epidermal growth factor receptor therapy improved outcome for KRAS wild-type CMS2 patients.
Conclusions: The CMS classification holds clear potential for clinical use in predicting both prognosis and response to systemic therapy, which seems to be independent of the classifier used. Prospective studies are warranted to support implementation of the CMS taxonomy in clinical practice.
© The Author(s) 2021. Published by Oxford University Press.
Figures

References
-
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. - PubMed
-
- Punt CJ, Koopman M, Vermeulen L. From tumour heterogeneity to advances in precision treatment of colorectal cancer. Nat Rev Clin Oncol. 2017;14(4):235–246. - PubMed
-
- Kopetz S, Grothey A, Yaeger R, et al. Encorafenib, binimetinib, and cetuximab in BRAF V600E-mutated colorectal cancer. N Engl J Med. 2019;381(17):1632–1643. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

