Health-related quality of life in patients with newly diagnosed multiple myeloma ineligible for stem cell transplantation: results from the randomized phase III ALCYONE trial
- PMID: 34078314
- PMCID: PMC8170980
- DOI: 10.1186/s12885-021-08325-2
Health-related quality of life in patients with newly diagnosed multiple myeloma ineligible for stem cell transplantation: results from the randomized phase III ALCYONE trial
Abstract
Background: In the phase III ALCYONE trial, daratumumab plus bortezomib/melphalan/prednisone (D-VMP) significantly improved overall response rate and progression-free status compared with VMP alone in transplant-ineligible patients with newly diagnosed multiple myeloma (NDMM). Here, we present patient-reported outcomes (PROs) from ALCYONE.
Methods: The European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30-item (EORTC QLQ-C30) and EuroQol 5-dimensional descriptive system (EQ-5D-5L) questionnaire were administered at baseline, every 3 months (year 1) and every 6 months (until progression). Treatment effects were assessed using a repeated-measures, mixed-effects model.
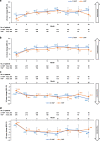
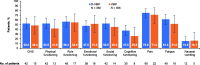
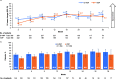
Results: Compliance with PRO assessments was comparable at baseline (> 90%) and throughout study (> 76%) for both treatment groups. Improvements from baseline were observed in both groups for EORTC QLQ-C30 Global Health Status (GHS), most functional scales, symptom scales and EQ-5D-5L visual analog scale (VAS). Between-group differences were significant for GHS (p = 0.0240) and VAS (p = 0.0160) at month 3. Improvements in pain were clinically meaningful in both groups at all assessment time points. Cognitive function declined in both groups, but the magnitude of the decline was not clinically meaningful.
Conclusions: Patients with transplant-ineligible NDMM demonstrated early and continuous improvements in health-related quality of life, including improvements in functioning and symptoms, following treatment with D-VMP or VMP.
Trial registration: ClinicalTrials.gov identifier NCT02195479 , registered September 21, 2014.
Conflict of interest statement
Stefan Knop received honoraria from Amgen, Bristol-Myers Squibb, Celgene, Janssen and Takeda; served as a consultant or in an advisory role for Amgen, Bristol-Myers Squibb, Celgene, Janssen and Takeda; and received research funding from Amgen, Bristol-Myers Squibb, Celgene, Janssen and Takeda. Maria-Victoria Mateos has received honoraria from Adaptive Biotechnologies, Amgen, Celgene, Janssen Pharmaceuticals and Takeda; served on the Board of Directors or advisory committees for AbbVie, Amgen, Celgene, GenTech, GlaxoSmithKline, Janssen Pharmaceuticals, Mundipharma, PharmaMar, Roche and Takeda; served on data and monitoring committees for Amgen and Janssen Pharmaceuticals; and served on speakers bureaus for Amgen, Celgene, Janssen Pharmaceuticals and Takeda. Meletios A. Dimopoulos has received consulting fees, honoraria and research funding from Amgen, Celgene, Janssen and Takeda; and participated in advisory boards for Sanofi-Aventis. Kenishi Suzuki received honoraria from Bristol-Myers Squibb, Celgene, Janssen, Novartis, Ono, Takeda and Sanofi-Aventis; served as a consultant or in an advisory role for Bristol-Myers Squibb, Celgene, Janssen, Novartis, Ono, Takeda and Sanofi-Aventis; and is employed by SRL. Andrzej Jakubowiak received honoraria from AbbVie, Adaptive Biotechnologies, Amgen, Bristol-Myers Squibb, Celgene, Janssen, Karyopharm, SkylineDx and Takeda; and served as a consultant or in an advisory role for AbbVie, Adaptive Biotechnologies, Amgen, Bristol-Myers Squibb, Celgene, Janssen, Karyopharm, SkylineDx and Takeda. Chantal Doyen served as a consultant or in an advisory role for Janssen. Paulo Lucio received honoraria from Amgen, Celgene, Janssen and Takeda; and served as a consultant or in an advisory role for Amgen, Celgene, Janssen and Takeda. Mark Cook received honoraria from AbbVie, Amgen, Bristol-Myers Squibb, Celgene, GlaxoSmithKline, Janssen and Takeda; served as a consultant or in an advisory role for AbbVie, Amgen, Bristol-Myers Squibb, Celgene, GlaxoSmithKline, Janssen and Takeda; received research support from Celgene and Janssen; and served on a speakers bureau for Celgene and Janssen. Sebastian Grosicki received research funding from Affimed. Sung-Soo Yoon served as a consultant or in an advisory role for Amgen, Astellas, Celgene, Chugai, Janssen, Novartis and Takeda; received honoraria from Novartis; and received research funding from Kyowa Kirin, Roche-Genentech and Yuhan Pharmaceutical. Mamta Garg has received honoraria from Amgen and Janssen; research funding from Novartis; and travel accommodations and expenses from Amgen, Novartis and Takeda. Jianping Wang, Susan Wroblewski, Anupa Kudva, Katharine S. Gries and John Fastenau are employed by Janssen. Jesus San-Miguel received honoraria from Amgen, Bristol-Myers Squibb, Celgene, Janssen, Novartis, Roche and Sanofi. Michele Cavo has consulted for AbbVie, Amgen, Celgene and Janssen Pharmaceuticals; received honoraria from AbbVie, Amgen, Bristol-Myers Squibb, Celgene, Janssen Pharmaceuticals and Takeda; and served on speakers bureaus for and received travel and/or accommodation expenses from Celgene and Janssen Pharmaceuticals. Zsolt Nagy, Ganna Usenko, Ludek Pour, Andre Crepaldi, Anna Marina Liberati, Philip Campbell, Tatiana Shelekhova, Genadi Iosava and Tomoaki Fujisaki have nothing to disclose.
Figures
References
-
- Moreau P, San Miguel J, Sonneveld P, Mateos MV, Zamagni E, Avet-Loiseau H, Hajek R, Dimopoulos MA, Ludwig H, Einsele H, et al. Multiple myeloma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28(suppl 4):iv52–iv61. doi: 10.1093/annonc/mdx096. - DOI - PubMed
-
- Rodriguez-Otero P, Mateos MV, Martinez-Lopez J, Hernandez MT, Ocio EM, Rosinol L, Martinez R, Teruel AI, Gutierrez NC, Bargay J, et al. Predicting long-term disease control in transplant-ineligible patients with multiple myeloma: impact of an MGUS-like signature. Blood Cancer J. 2019;9(4):36. doi: 10.1038/s41408-019-0176-x. - DOI - PMC - PubMed
-
- Gandolfi S, Vekstein C, Laubach JP, O'Brien A, Masone K, Munshi NC, Anderson KC, Richardson PG. The evolving role of transplantation in multiple myeloma: the need for a heterogeneous approach to a heterogeneous disease. Clin Adv Hematol Oncol. 2018;16(8):564–574. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

