Direct and indirect costs and cost-driving factors in adults with tuberous sclerosis complex: a multicenter cohort study and a review of the literature
- PMID: 34078440
- PMCID: PMC8170458
- DOI: 10.1186/s13023-021-01838-w
Direct and indirect costs and cost-driving factors in adults with tuberous sclerosis complex: a multicenter cohort study and a review of the literature
Abstract
Background: Tuberous sclerosis complex (TSC) is a monogenetic, multisystem disorder characterized by benign growths due to TSC1 or TSC2 mutations. This German multicenter study estimated the costs and related cost drivers associated with organ manifestations in adults with TSC.
Methods: A validated, three-month, retrospective questionnaire assessed the sociodemographic and clinical characteristics, organ manifestations, direct, indirect, out-of-pocket (OOP), and nursing care-level costs among adult individuals with TSC throughout Germany from a societal perspective (costing year: 2019).
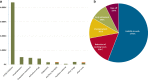
Results: We enrolled 192 adults with TSC (mean age: 33.4 ± 12.7 years; range: 18-78 years, 51.6% [n = 99] women). Reported TSC disease manifestations included skin (94.8%) and kidney and urinary tract (74%) disorders, epilepsy (72.9%), structural brain defects (67.2%), psychiatric disorders (50.5%), heart and circulatory system disorders (50.5%), and lymphangioleiomyomatosis (11.5%). TSC1 and TSC2 mutations were reported in 16.7% and 25% of respondents, respectively. Mean direct health care costs totaled EUR 6452 (median EUR 1920; 95% confidence interval [CI] EUR 5533-7422) per patient over three months. Medication costs represented the major direct cost category (77% of total direct costs; mean EUR 4953), and mechanistic target of rapamycin (mTOR) inhibitors represented the largest share (68%, EUR 4358). Mean antiseizure drug (ASD) costs were only EUR 415 (6%). Inpatient costs (8%, EUR 518) and outpatient treatment costs (7%; EUR 467) were important further direct cost components. The mean care grade allowance as an approximator of informal nursing care costs was EUR 929 (median EUR 0; 95% CI EUR 780-1083) over three months. Mean indirect costs totaled EUR 3174 (median EUR 0; 95% CI EUR 2503-3840) among working-age individuals (< 67 years in Germany). Multiple regression analyses revealed mTOR inhibitor use and persistent seizures as independent cost-driving factors for total direct costs. Older age and disability were independent cost-driving factors for total indirect costs, whereas epilepsy, psychiatric disease, and disability were independent cost-driving factors for nursing care costs.
Conclusions: This three-month study revealed substantial direct healthcare, indirect healthcare, and medication costs associated with TSC in Germany. This study highlights the spectrum of organ manifestations and their associated treatment needs in the German healthcare setting.
Trial registration: DRKS, DRKS00016045. Registered 01 March 2019, http://www.drks.de/DRKS00016045 .
Keywords: Angiomyolipoma; Anticonvulsant; Costs; Epilepsy; Genetics; MTOR inhibitor; Seizure; Sociodemographic characteristics; Subependymal giant cell astrocytoma; TSC.
Conflict of interest statement
JPZ reports speakers’ honoraria and travel grants from EISAI and Desitin Arzneimittel. FR reports personal fees from Arvelle Therapeutics, Desitin Arzneimittel, Eisai, GW Pharmaceuticals companies, Novartis, Medtronic, Sandoz, Shire, and UCB, and grants from the Detlev-Wrobel-Fonds for Epilepsy Research, the Deutsche Forschungsgemeinschaft, the LOEWE Programme of the State of Hesse, and the European Union. MS reports personal fees from Novartis and GW Pharmaceuticals companies. GK reports personal fees from Desitin Arzneimittel, Eisai, GW Pharmaceuticals companies, UCB, Novartis, Takeda, and Zogenix. TM reports personal fees and grants from Arvelle Therapeutics, Eisai, GW Pharmaceuticals companies, UCB, and Zogenix. AB reports personal fees from Desitin Arzneimittel GmbH, Eisai GmbH, Eisai GmbH, Shire GmbH, UCB Pharma GmbH, and ViroPharma GmbH. KMK reports personal fees from UCB Pharma, Novartis Pharma AG, Eisai, and GW Pharmaceuticals, grants from the federal state Hessen through the LOEWE program, and from the Canadian Institutes of Health Research. SM reports grants from Novartis, UCB, Shire, Deutsche Forschungsgemeinschaft and Epilepsiestiftung Dr. Wolf. FvP reports personal fees and grants from Bial, Desitin Arzneimittel, Eisai, GW Pharmaceutical companies, Arvelle Therapeutics, and UCB Pharma. SS-B reports personal fees from Eisai, Desitin Pharma, GW Pharmaceuticals companies, LivaNova, UCB, and Zogenix. AS reports personal fees and grants from Arvelle Therapeutics, Desitin Arzneimittel, Eisai, GW Pharmaceuticals companies, LivaNova, Marinus Pharma, Medtronic, UCB, and Zogenix. None of the other authors reported any related conflicts of interest.
Figures
References
-
- Ebrahimi-Fakhari D, Mann LL, Poryo M, Graf N, von Kries R, Heinrich B, Ebrahimi-Fakhari D, Flotats-Bastardas M, Gortner L, Zemlin M, et al. Incidence of tuberous sclerosis and age at first diagnosis: new data and emerging trends from a national, prospective surveillance study. Orphanet. J. Rare Dis. 2018;13(1):117. doi: 10.1186/s13023-018-0870-y. - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous