Steroid Hormone Biosynthesis Metabolism Is Associated With Fatigue Related to Androgen Deprivation Therapy for Prostate Cancer
- PMID: 34079794
- PMCID: PMC8166231
- DOI: 10.3389/fcell.2021.642307
Steroid Hormone Biosynthesis Metabolism Is Associated With Fatigue Related to Androgen Deprivation Therapy for Prostate Cancer
Abstract
Background: Androgen deprivation therapy (ADT) is a cornerstone treatment for prostate cancer. Despite the clinical benefits, ADT is associated with multiple adverse effects including fatigue. The goal of the study was to examine metabolomic changes to better understand cancer-related fatigue specific to ADT treatment.
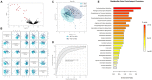
Methods: A total of 160 plasma samples collected from participants with (+ADT, n = 58) or without neoadjuvant ADT (-ADT, n = 102) prior to radiation therapy for treatment of non-metastatic localized prostate cancer were included in the study. Fatigue and sleep-related impairment were measured using the Patient Reported Outcomes Measurement Information System. Plasma metabolites were identified and measured using untargeted ultrahigh-performance liquid chromatography/mass spectrometry metabolomics analyses. Partial least square discriminant analysis was used to identify discriminant metabolite features, and the diagnostic performance of selected classifiers was quantified using AUROC curve analysis. Pathway enrichment analysis was performed using metabolite sets enrichment analyses.
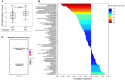
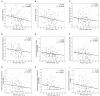
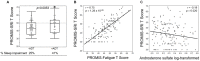
Findings: Steroid hormone biosynthesis pathways, including androstenedione metabolism as well as androgen and estrogen metabolism, were overrepresented by metabolites that significantly discriminated samples in the +ADT from the -ADT group. Additional overrepresented metabolic pathways included amino acid metabolism, glutathione metabolism, and carnitine synthesis. Of the metabolites that were significantly different between the groups, steroid hormone biosynthesis metabolites were most significantly correlated with fatigue severity. Sleep-related impairment was strongly correlated with fatigue severity and inversely correlated with ADT-induced reduction in androsterone sulfate.
Conclusions: Patients with non-metastatic prostate cancer receiving neoadjuvant ADT prior to radiation therapy reported relatively more severe fatigue. Increased fatigue in this population may be attributable to sleep-related impairment associated with alterations in steroid hormone biosynthesis. Findings in this study provide a basis for further research of changes in sleep patterns and their role in this specific subcategory of cancer-related fatigue caused by the treatment.
Keywords: androgen deprivation therapy; androgen metabolism; cancer-related fatigue; metabolomics; prostate cancer; radiation therapy; steroid hormone biosynthesis.
Copyright © 2021 Feng, Barb, Allen, Regan and Saligan.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Plasma metabolomic profile associated with fatigue in cancer patients.Cancer Med. 2021 Mar;10(5):1623-1633. doi: 10.1002/cam4.3749. Epub 2021 Feb 3. Cancer Med. 2021. PMID: 33534943 Free PMC article.
-
Metabolomic effects of androgen deprivation therapy treatment for prostate cancer.Cancer Med. 2020 Jun;9(11):3691-3702. doi: 10.1002/cam4.3016. Epub 2020 Mar 31. Cancer Med. 2020. PMID: 32232974 Free PMC article. Clinical Trial.
-
Quality of sleep in patients receiving androgen deprivation therapy for prostate cancer.Neurol Sci. 2017 Aug;38(8):1445-1451. doi: 10.1007/s10072-017-2989-3. Epub 2017 May 8. Neurol Sci. 2017. PMID: 28484881
-
Androgen deprivation therapy for prostate cancer: long-term safety and patient outcomes.Patient Relat Outcome Meas. 2014 Jul 5;5:63-70. doi: 10.2147/PROM.S52788. eCollection 2014. Patient Relat Outcome Meas. 2014. PMID: 25045284 Free PMC article. Review.
-
Intermittent androgen deprivation therapy in advanced prostate cancer.Curr Treat Options Oncol. 2014 Mar;15(1):127-36. doi: 10.1007/s11864-013-0272-2. Curr Treat Options Oncol. 2014. PMID: 24395278 Review.
Cited by
-
A Molecular Approach to Understanding the Role of Diet in Cancer-Related Fatigue: Challenges and Future Opportunities.Nutrients. 2022 Apr 2;14(7):1496. doi: 10.3390/nu14071496. Nutrients. 2022. PMID: 35406105 Free PMC article. Review.
-
Causality of genetically determined metabolites on anxiety disorders: a two-sample Mendelian randomization study.J Transl Med. 2022 Oct 20;20(1):475. doi: 10.1186/s12967-022-03691-2. J Transl Med. 2022. PMID: 36266699 Free PMC article.
-
Luteinizing hormone-releasing hormone receptor agonists and antagonists in prostate cancer: effects on long-term survival and combined therapy with next-generation hormonal agents.Cancer Biol Med. 2024 Dec 24;21(11):1012-32. doi: 10.20892/j.issn.2095-3941.2024.0139. Cancer Biol Med. 2024. PMID: 39718161 Free PMC article. Review.
-
Metabolic underpinnings of cancer-related fatigue.Am J Physiol Endocrinol Metab. 2024 Mar 1;326(3):E290-E307. doi: 10.1152/ajpendo.00378.2023. Epub 2024 Jan 31. Am J Physiol Endocrinol Metab. 2024. PMID: 38294698 Free PMC article. Review.
-
Understanding the mechanism behind preoperative exercise therapy in patients with gastrointestinal cancers: a prospective randomized clinical trial.BMC Sports Sci Med Rehabil. 2025 Mar 14;17(1):50. doi: 10.1186/s13102-025-01094-6. BMC Sports Sci Med Rehabil. 2025. PMID: 40087747 Free PMC article.
References
-
- Ameringer S., Elswick R. K., Jr., Menzies V., Robins J. L., Starkweather A., Walter J., et al. (2016). Psychometric evaluation of the patient-reported outcomes measurement information system fatigue-short form across diverse populations. Nurs. Res. 65 279–289. 10.1097/NNR.0000000000000162 - DOI - PMC - PubMed
-
- Benjamini Y., Hochberg Y. (1995). Controlling the false discovery rate – a practical and powerful Mpproach to multiple testing. J. R. Stat. Soc. Ser. B Stat. Methodol. 57 289–300.
LinkOut - more resources
Full Text Sources

