Synthesis of 4,4'-(arylmethylene)bis(3-methyl-1-phenyl-1H-pyrazol-5-ols) and evaluation of their antioxidant and anticancer activities
- PMID: 34082794
- PMCID: PMC8176600
- DOI: 10.1186/s13065-021-00765-y
Synthesis of 4,4'-(arylmethylene)bis(3-methyl-1-phenyl-1H-pyrazol-5-ols) and evaluation of their antioxidant and anticancer activities
Abstract
Background: Pyrazoles have attracted particular attention due to the diverse biological activities associated with this heterocyclic system, and some have been shown to be cytotoxic to several human cell lines. Several drugs currently on the market have this heterocycle as the key structural motif, and some have been approved for the treatment of different types of cancer.
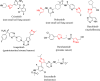
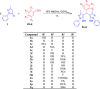
Results: 4,4'-(Arylmethylene)bis(1H-pyrazol-5-ols) derivatives 3a-q were synthetized by a three components reaction of 3-methyl-1-phenyl-5-pyrazolone (1) with various benzaldehydes 2 catalyzed by sodium acetate at room temperature. The structures of all synthesized compounds were characterized by physicochemical properties and spectral means (IR and NMR) and were evaluated for their radical scavenging activity by DPPH assay and tested in vitro on colorectal RKO carcinoma cells in order to determine their cytotoxic properties. All 4,4'-(arylmethylene)bis(1H-pyrazol-5-ols) derivatives 3a-q were synthetized in high to excellent yield, and pure products were isolated by simple filtration. All compounds have good radical scavenging activity, and half of them are more active than ascorbic acid used as standard.
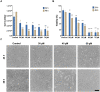
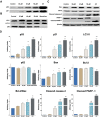
Conclusion: Several derivatives proved to be cytotoxic in the RKO cell line. In particular, compound 3i proved to be a very potent scavenger with an IC50 of 6.2 ± 0.6 µM and exhibited an IC50 of 9.9 ± 1.1 μM against RKO cell. Autophagy proteins were activated as a survival mechanism, whereas the predominant pathway of death was p53-mediated apoptosis.
Keywords: 4,4ʹ-(arylmethylene)bis(1H-pyrazol-5-ols); Antioxidant; Apoptosis; Autophagy.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Similar articles
-
Visible Light-Promoted Green and Sustainable Approach for One-Pot Synthesis of 4,4'-(Arylmethylene)bis(1H-pyrazol-5-ols), In Vitro Anticancer Activity, and Molecular Docking with Covid-19 Mpro.ACS Omega. 2022 Sep 14;7(38):34583-34598. doi: 10.1021/acsomega.2c04506. eCollection 2022 Sep 27. ACS Omega. 2022. PMID: 36188265 Free PMC article.
-
Evaluation of the Anti-Leishmania mexicana and -Trypanosoma brucei Activity and Mode of Action of 4,4'-(Arylmethylene)bis(3-methyl-1-phenyl-1H-pyrazol-5-ol).Biomedicines. 2022 Aug 7;10(8):1913. doi: 10.3390/biomedicines10081913. Biomedicines. 2022. PMID: 36009460 Free PMC article.
-
Poly(aniline-co-melamine)@MnFe2O4 nanocatalyst for the synthesis of 4,4'-(arylmethylene) bis (1H-pyrazole-5-ol) derivatives, and 1,4- dihydropyrano[2,3-c]pyrazoles and evaluation of their antioxidant, and anticancer activities.Front Chem. 2022 Oct 28;10:1046120. doi: 10.3389/fchem.2022.1046120. eCollection 2022. Front Chem. 2022. PMID: 36385997 Free PMC article.
-
Synthesis and antiviral activity of 4,4'-(arylmethylene)bis(1H-pyrazol-5-ols) against peste des petits ruminant virus (PPRV).Bioorg Med Chem Lett. 2009 Aug 1;19(15):4501-3. doi: 10.1016/j.bmcl.2009.02.113. Epub 2009 Mar 3. Bioorg Med Chem Lett. 2009. PMID: 19482473
-
Synthesis, Characterization, Antimicrobial Activity and Anticancer of Some New Pyrazolo[1,5-a]pyrimidines and Pyrazolo[5,1-c]1,2,4-triazines.Med Chem. 2020;16(6):750-760. doi: 10.2174/1573406415666190620144404. Med Chem. 2020. PMID: 31218963
Cited by
-
Pyrazolone-type compounds (part II): in vitro and in silico evaluation of antioxidant potential; structure-activity relationship.RSC Adv. 2023 Jan 18;13(5):2884-2895. doi: 10.1039/d2ra08280b. eCollection 2023 Jan 18. RSC Adv. 2023. PMID: 36756409 Free PMC article.
-
Visible Light-Promoted Green and Sustainable Approach for One-Pot Synthesis of 4,4'-(Arylmethylene)bis(1H-pyrazol-5-ols), In Vitro Anticancer Activity, and Molecular Docking with Covid-19 Mpro.ACS Omega. 2022 Sep 14;7(38):34583-34598. doi: 10.1021/acsomega.2c04506. eCollection 2022 Sep 27. ACS Omega. 2022. PMID: 36188265 Free PMC article.
-
Exploring the Multifaceted Biological Activities of Anthocyanins Isolated from Two Andean Berries.Foods. 2024 Aug 21;13(16):2625. doi: 10.3390/foods13162625. Foods. 2024. PMID: 39200552 Free PMC article.
-
Multicomponent synthesis of pyrano-pyrazolo-pyridones by a bimetallic cobalt-cadmium magnetic catalyst.Sci Rep. 2025 Aug 12;15(1):29462. doi: 10.1038/s41598-025-15535-2. Sci Rep. 2025. PMID: 40790353 Free PMC article.
-
Evaluation of the Anti-Leishmania mexicana and -Trypanosoma brucei Activity and Mode of Action of 4,4'-(Arylmethylene)bis(3-methyl-1-phenyl-1H-pyrazol-5-ol).Biomedicines. 2022 Aug 7;10(8):1913. doi: 10.3390/biomedicines10081913. Biomedicines. 2022. PMID: 36009460 Free PMC article.
References
-
- Gomtsyan A. Heterocycles in drugs and drug discovery. Chem Heterocycl Compd. 2012;48:7–10. doi: 10.1007/s10593-012-0960-z. - DOI
-
- Pandey A, Dewangan D, Verma S, et al. Synthesis of Schiff bases of 2-amino-5-aryl-1, 3, 4-thiadiazole And its Analgesic, Anti- Inflammatory, Anti-Bacterial and Anti- Tubercular Activity. Int J ChemTech Res. 2011;3:178–184.
-
- Halim KNM, Ramadan SK, Rizk SA, El-Hashash MA. Synthesis, DFT study, molecular docking and insecticidal evaluation of some pyrazole-based tetrahydropyrimidine derivatives. Synth Commun. 2020;50:1159–1175. doi: 10.1080/00397911.2020.1720739. - DOI
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
