Establishment of an integrated automated embryonic manipulation system for producing genetically modified mice
- PMID: 34083640
- PMCID: PMC8175685
- DOI: 10.1038/s41598-021-91148-9
Establishment of an integrated automated embryonic manipulation system for producing genetically modified mice
Abstract
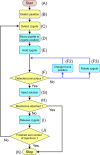
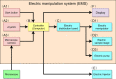
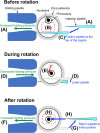
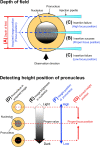
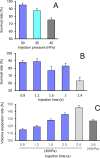
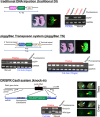
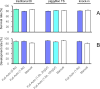
Genetically modified mice are commonly used in biologic, medical, and drug discovery research, but conventional microinjection methods used for genetic modification require extensive training and practical experience. Here we present a fully automated system for microinjection into the pronucleus to facilitate genetic modification. We first developed software that automatically controls the microinjection system hardware. The software permits automatic rotation of the zygote to move the pronucleus to the injection pipette insertion position. We also developed software that recognizes the pronucleus in 3-dimensional coordinates so that the injection pipette can be automatically inserted into the pronucleus, and achieved a 94% insertion rate by linking the 2 pieces of software. Next, we determined the optimal solution injection conditions (30 hPa, 0.8-2.0 s) by examining the survival rate of injected zygotes. Finally, we produced transgenic (traditional DNA injection and piggyBac Transposon system) and knock-in (genomic editing) mice using our newly developed Integrated Automated Embryo Manipulation System (IAEMS). We propose that the IAEMS will simplify highly reproducible pronuclear stage zygote microinjection procedures.
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Nanoinjection: pronuclear DNA delivery using a charged lance.Transgenic Res. 2012 Dec;21(6):1279-90. doi: 10.1007/s11248-012-9610-6. Epub 2012 Mar 14. Transgenic Res. 2012. PMID: 22415347
-
Pronuclear Microinjection of One-Cell Embryos.Methods Mol Biol. 2020;2066:27-33. doi: 10.1007/978-1-4939-9837-1_2. Methods Mol Biol. 2020. PMID: 31512204
-
Efficiency of transgenic rat production is independent of transgene-construct and overnight embryo culture.Theriogenology. 2004 May;61(7-8):1441-53. doi: 10.1016/j.theriogenology.2003.08.006. Theriogenology. 2004. PMID: 15036975
-
Challenges and progress in the production of transgenic cattle.Reprod Fertil Dev. 1994;6(5):647-52. doi: 10.1071/rd9940647. Reprod Fertil Dev. 1994. PMID: 7569045 Review.
-
Generation of Mammalian offspring by haploid embryonic stem cells microinjection.Curr Protoc Stem Cell Biol. 2014 Nov 3;31:1A.6.1-15. doi: 10.1002/9780470151808.sc01a06s31. Curr Protoc Stem Cell Biol. 2014. PMID: 25366896 Review.
Cited by
-
Genome editing.Sci Rep. 2022 Nov 28;12(1):20497. doi: 10.1038/s41598-022-24850-x. Sci Rep. 2022. PMID: 36443399 Free PMC article.
-
Basic Principles of RNA Interference: Nucleic Acid Types and In Vitro Intracellular Delivery Methods.Micromachines (Basel). 2023 Jun 27;14(7):1321. doi: 10.3390/mi14071321. Micromachines (Basel). 2023. PMID: 37512632 Free PMC article. Review.
References
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials

