A Complex of LaoA and LaoB Acts as a Tat-Dependent Dehydrogenase for Long-Chain Alcohols in Pseudomonas aeruginosa
- PMID: 34085859
- PMCID: PMC8315182
- DOI: 10.1128/AEM.00762-21
A Complex of LaoA and LaoB Acts as a Tat-Dependent Dehydrogenase for Long-Chain Alcohols in Pseudomonas aeruginosa
Abstract
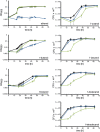
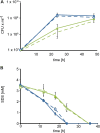
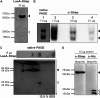
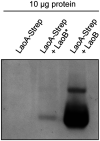
The opportunistic pathogen Pseudomonas aeruginosa can utilize unusual carbon sources, like sodium dodecyl sulfate (SDS) and alkanes. Whereas the initiating enzymatic steps of the corresponding degradation pathways have been characterized in detail, the oxidation of the emerging long-chain alcohols has received little attention. Recently, the genes for the Lao (
Keywords: Pseudomonas aeruginosa; SDS degradation; Tat system; alcohol dehydrogenase; cytochrome c; flavin-dependent alcohol dehydrogenase; flavoenzymes; hitchhiker mechanism; long-chain-alcohol oxidation; protein complex; rhamnolipids.
Figures

Similar articles
-
LaoABCR, a Novel System for Oxidation of Long-Chain Alcohols Derived from SDS and Alkane Degradation in Pseudomonas aeruginosa.Appl Environ Microbiol. 2018 Jun 18;84(13):e00626-18. doi: 10.1128/AEM.00626-18. Print 2018 Jul 1. Appl Environ Microbiol. 2018. PMID: 29678916 Free PMC article.
-
Sulfate Ester Detergent Degradation in Pseudomonas aeruginosa Is Subject to both Positive and Negative Regulation.Appl Environ Microbiol. 2019 Nov 14;85(23):e01352-19. doi: 10.1128/AEM.01352-19. Print 2019 Dec 1. Appl Environ Microbiol. 2019. PMID: 31540990 Free PMC article.
-
A soluble two-component regulatory system controls expression of quinoprotein ethanol dehydrogenase (QEDH) but not expression of cytochrome c(550) of the ethanol-oxidation system in Pseudomonas aeruginosa.Microbiology (Reading). 2001 Feb;147(Pt 2):363-372. doi: 10.1099/00221287-147-2-363. Microbiology (Reading). 2001. PMID: 11158353
-
Hydride transfer made easy in the reaction of alcohol oxidation catalyzed by flavin-dependent oxidases.Biochemistry. 2008 Dec 30;47(52):13745-53. doi: 10.1021/bi801994c. Biochemistry. 2008. PMID: 19053234 Review.
-
The ethanol oxidation system and its regulation in Pseudomonas aeruginosa.Biochim Biophys Acta. 2003 Apr 11;1647(1-2):98-102. doi: 10.1016/s1570-9639(03)00066-9. Biochim Biophys Acta. 2003. PMID: 12686116 Review.
Cited by
-
A Plasmid-Borne Gene Cluster Flanked by Two Restriction-Modification Systems Enables an Arctic Strain of Psychrobacter sp. to Decompose SDS.Int J Mol Sci. 2023 Dec 31;25(1):551. doi: 10.3390/ijms25010551. Int J Mol Sci. 2023. PMID: 38203722 Free PMC article.
References
-
- Thomas OR, White GF. 1989. Metabolic pathway for the biodegradation of sodium dodecyl sulfate by Pseudomonas sp. C12B. Biotechnol Appl Biochem 11:318–327. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

