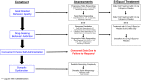
S-Equol mitigates motivational deficits and dysregulation associated with HIV-1
- PMID: 34088932
- PMCID: PMC8178385
- DOI: 10.1038/s41598-021-91240-0
S-Equol mitigates motivational deficits and dysregulation associated with HIV-1
Abstract
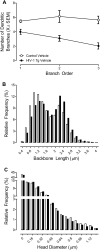
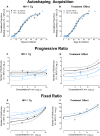
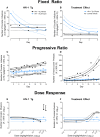
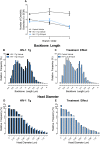
Motivational deficits (e.g., apathy) and dysregulation (e.g., addiction) in HIV-1 seropositive individuals, despite treatment with combination antiretroviral therapy, necessitates the development of innovative adjunctive therapeutics. S-Equol (SE), a selective estrogen receptor β agonist, has been implicated as a neuroprotective and/or neurorestorative therapeutic for HIV-1 associated neurocognitive disorders (HAND); its therapeutic utility for motivational alterations, however, has yet to be systematically evaluated. Thus, HIV-1 transgenic (Tg) and control animals were treated with either a daily oral dose of SE (0.2 mg) or vehicle and assessed in a series of tasks to evaluate goal-directed and drug-seeking behavior. First, at the genotypic level, motivational deficits in HIV-1 Tg rats treated with vehicle were characterized by a diminished reinforcing efficacy of, and sensitivity to, sucrose. Motivational dysregulation was evidenced by enhanced drug-seeking for cocaine relative to control animals treated with vehicle. Second, treatment with SE ameliorated both motivational deficits and dysregulation in HIV-1 Tg rats. Following a history of cocaine self-administration, HIV-1 Tg animals treated with vehicle exhibited lower levels of dendritic branching and a shift towards longer dendritic spines with decreased head diameter. Treatment with SE, however, led to long-term enhancements in dendritic spine morphology in HIV-1 Tg animals supporting a potential underlying basis by which SE exerts its therapeutic effects. Taken together, SE restored motivated behavior in the HIV-1 Tg rat, expanding the potential clinical utility of SE to include both neurocognitive and affective alterations.
Conflict of interest statement
The authors declare no competing interests.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

