Efficacy and Safety of Medication for Attention-Deficit Hyperactivity Disorder in Children and Adolescents with Common Comorbidities: A Systematic Review
- PMID: 34089145
- PMCID: PMC8571469
- DOI: 10.1007/s40120-021-00249-0
Efficacy and Safety of Medication for Attention-Deficit Hyperactivity Disorder in Children and Adolescents with Common Comorbidities: A Systematic Review
Abstract
Introduction: Comorbid psychiatric conditions in children and adolescents with attention-deficit hyperactivity disorder (ADHD) occur frequently, complicate management, and are associated with substantial burden on patients and caregivers. Very few systematic reviews have assessed the efficacy and safety of medications for ADHD in children and adolescents with comorbidities. Of those that were conducted, most focused on a particular comorbidity or medication. In this systematic literature review, we summarize the efficacy and safety of treatments for children and adolescents with ADHD and comorbid autism spectrum disorders, oppositional defiant disorder, Tourette's disorder and other tic disorders, generalized anxiety disorder, and major depressive disorder.
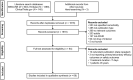
Methods: We searched MEDLINE, Embase, and ClinicalTrials.gov (to October 2019) for studies of patients (aged < 18 years) with an ADHD diagnosis and the specified comorbidities treated with amphetamines, methylphenidate and derivatives, atomoxetine (ATX), and guanfacine extended-release (GXR). For efficacy, placebo-controlled randomized controlled trials (RCTs) or meta-analyses of RCTs were eligible for inclusion; for safety, all study types were eligible. The primary efficacy outcome measure was ADHD Rating Scale IV (ADHD-RS-IV) total score.
Results: Of 2177 publications/trials retrieved, 69 were included in this systematic literature review (5 meta-analyses, 37 placebo-controlled RCTs, 16 cohort studies, 11 case reports). A systematic narrative synthesis is provided because insufficient data were retrieved to combine ADHD-RS-IV total scores or effect sizes. Effect sizes for ADHD-RS-IV total scores were available for ten RCTs and ranged from 0.46 to 1.0 for ATX and from 0.92 to 2.0 for GXR across comorbidities. The numbers and types of adverse events in children with comorbidities were consistent with those in children without comorbidities, but treatment should be individualized to ensure children can tolerate the lowest effective dose.
Conclusion: Limited information is available from placebo-controlled RCTs on the efficacy (by ADHD-RS-IV) or safety of medication in children with ADHD and psychiatric comorbidities. Further studies are required to support evidence-based drug selection for these populations.
Keywords: Adolescent; Attention deficit hyperactivity disorder; Child; Comorbidity; Pharmacotherapies.
© 2021. The Author(s).
Figures

References
-
- Jensen CM, Steinhausen HC. Comorbid mental disorders in children and adolescents with attention-deficit/hyperactivity disorder in a large nationwide study. Atten Defic Hyperact Disord. 2015;7(1):27–38. - PubMed
-
- Reale L, Bartoli B, Cartabia M, et al. Comorbidity prevalence and treatment outcome in children and adolescents with ADHD. Eur Child Adolesc Psychiatry. 2017;26(12):1443–1457. - PubMed
-
- Klassen AF, Miller A, Fine S. Health-related quality of life in children and adolescents who have a diagnosis of attention-deficit/hyperactivity disorder. Pediatrics. 2004;114(5):e541–e547. - PubMed
-
- Sparrow EP, Erhardt D. Essentials of ADHD assessment for children and adolescents. In: Kaufman AS, Kaufman NL, series editors. Essentials of psychological assessment. Hoboken: Wiley; 2014.
Publication types
LinkOut - more resources
Full Text Sources

