Gatekeeper function for Short stop at the ring canals of the Drosophila ovary
- PMID: 34089646
- PMCID: PMC8355207
- DOI: 10.1016/j.cub.2021.05.010
Gatekeeper function for Short stop at the ring canals of the Drosophila ovary
Abstract
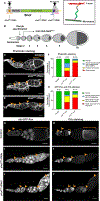
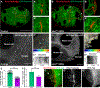
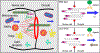
Growth of the Drosophila oocyte requires transport of cytoplasmic materials from the interconnected sister cells (nurse cells) through ring canals, the cytoplasmic bridges that remained open after incomplete germ cell division. Given the open nature of the ring canals, it is unclear how the direction of transport through the ring canal is controlled. In this work, we show that a single Drosophila spectraplakin Short stop (Shot) controls the direction of flow from nurse cells to the oocyte. Knockdown of shot changes the direction of transport through the ring canals from unidirectional (toward the oocyte) to bidirectional. After shot knockdown, the oocyte stops growing, resulting in a characteristic small oocyte phenotype. In agreement with this transport-directing function of Shot, we find that it is localized at the asymmetric actin baskets on the nurse cell side of the ring canals. In wild-type egg chambers, microtubules localized in the ring canals have uniform polarity (minus ends toward the oocyte), while in the absence of Shot, these microtubules have mixed polarity. Together, we propose that Shot functions as a gatekeeper directing transport from nurse cells to the oocyte via the organization of microtubule tracks to facilitate the transport driven by the minus-end-directed microtubule motor cytoplasmic dynein. VIDEO ABSTRACT.
Keywords: Drosophila; actin; cytoskeleton; dynein; intercellular transport; microtubules; molecular motor; oocyte; polarity; spectraplakin.
Copyright © 2021 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures



Comment in
-
Cell biology: Short stop is a team player in intercellular transport.Curr Biol. 2021 Aug 9;31(15):R959-R962. doi: 10.1016/j.cub.2021.06.042. Curr Biol. 2021. PMID: 34375601
References
-
- Roper K, Gregory SL, and Brown NH (2002). The ‘Spectraplakins’: cytoskeletal giants with characteristics of both spectrin and plakin families. Journal of cell science 115, 4215–4225. - PubMed
-
- Alves-Silva J, Sanchez-Soriano N, Beaven R, Klein M, Parkin J, Millard TH, Bellen HJ, Venken KJ, Ballestrem C, Kammerer RA, et al. (2012). Spectraplakins promote microtubule-mediated axonal growth by functioning as structural microtubule-associated proteins and EB1-dependent +TIPs (tip interacting proteins). The Journal of neuroscience : the official journal of the Society for Neuroscience 32, 9143–9158. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

