Lack of detectable short-term effects of a single dose of ivermectin on the human immune system
- PMID: 34090504
- PMCID: PMC8179708
- DOI: 10.1186/s13071-021-04810-6
Lack of detectable short-term effects of a single dose of ivermectin on the human immune system
Abstract
Background: Ivermectin is widely used in human and animal medicine to treat and prevent parasite nematode infections. It has been suggested that its mode of action requires the host immune system, as it is difficult to reproduce its clinical efficacy in vitro. We therefore studied the effects of a single dose of ivermectin (Stromectol®-0.15 mg/kg) on cytokine levels and immune cell gene expression in human volunteers. This dose reduces bloodstream microfilariae rapidly and for several months when given in mass drug administration programmes.
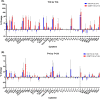
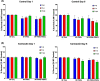
Methods: Healthy volunteers with no travel history to endemic regions were given 3-4 tablets, depending on their weight, of either ivermectin or a placebo. Blood samples were drawn immediately prior to administration, 4 h and 24 h afterwards, and complete blood counts performed. Serum levels of 41 cytokines and chemokines were measured using Luminex® and expression levels of 770 myeloid-cell-related genes determined using the NanoString nCounter®. Cytokine levels at 4 h and 24 h post-treatment were compared to the levels pre-treatment using simple t tests to determine if any individual results required further investigation, taking p = < 0.05 as the level of significance. NanoString data were analysed on the proprietary software, nSolver™.
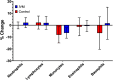
Results: No significant differences were observed in complete blood counts or cytokine levels at either time point between people given ivermectin versus placebo. Only three genes showed a significant change in expression in peripheral blood mononuclear cells 4 h after ivermectin was given; there were no significant changes 24 h after drug administration or in polymorphonuclear cells at either time point. Leukocytes isolated from those participants given ivermectin showed no difference in their ability to kill Brugia malayi microfilariae in vitro.
Conclusions: Overall, our data do not support a direct effect of ivermectin, when given at the dose used in current filarial elimination programmes, on the human immune system. Trial registration ClinicalTrials.gov NCT03459794 Registered 9th March 2018, Retrospectively registered https://clinicaltrials.gov/ct2/show/NCT03459794?term=NCT03459794&draw=2&rank=1 .
Keywords: Cytokine; Ivermectin; Lymphatic filariasis; Peripheral blood mononuclear cells; Polymorphonuclear cells.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

Similar articles
-
Recognition and killing of Brugia malayi microfilariae by human immune cells is dependent on the parasite sample and is not altered by ivermectin treatment.Int J Parasitol Drugs Drug Resist. 2018 Dec;8(3):587-595. doi: 10.1016/j.ijpddr.2018.09.002. Epub 2018 Sep 22. Int J Parasitol Drugs Drug Resist. 2018. PMID: 30279092 Free PMC article.
-
Ivermectin-dependent attachment of neutrophils and peripheral blood mononuclear cells to Dirofilaria immitis microfilariae in vitro.Vet Parasitol. 2014 Nov 15;206(1-2):38-42. doi: 10.1016/j.vetpar.2014.02.004. Epub 2014 Feb 12. Vet Parasitol. 2014. PMID: 24594213
-
Therapeutic trial of doxycyclin plus ivermectin for the treatment of Brugia malayi naturally infected cats.Vet Parasitol. 2017 Oct 15;245:42-47. doi: 10.1016/j.vetpar.2017.08.009. Epub 2017 Aug 18. Vet Parasitol. 2017. PMID: 28969836
-
The role of albendazole in programmes to eliminate lymphatic filariasis.Parasitol Today. 1999 Sep;15(9):382-6. doi: 10.1016/s0169-4758(99)01486-6. Parasitol Today. 1999. PMID: 10461168 Review.
-
Model-based analysis of trial data: microfilaria and worm-productivity loss after diethylcarbamazine-albendazole or ivermectin-albendazole combination therapy against Wuchereria bancrofti.Trop Med Int Health. 2006 May;11(5):718-28. doi: 10.1111/j.1365-3156.2006.01606.x. Trop Med Int Health. 2006. PMID: 16640625 Review.
References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

