Enantioselective Kinetic Resolution/Desymmetrization of Para-Quinols: A Case Study in Boronic-Acid-Directed Phosphoric Acid Catalysis
- PMID: 34093103
- PMCID: PMC8174618
- DOI: 10.1002/adsc.201900816
Enantioselective Kinetic Resolution/Desymmetrization of Para-Quinols: A Case Study in Boronic-Acid-Directed Phosphoric Acid Catalysis
Abstract
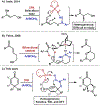
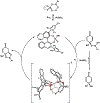
A chiral phosphoric acid-catalyzed kinetic resolution and desymmetrization of para-quinols operating via oxa-Michael addition was developed and subsequently subjected to mechanistic study. Good to excellent s-factors/enantioselectivities were obtained over a broad range of substrates. Kinetic studies were performed, and DFT studies favor a hydrogen bonding activation mode. The mechanistic studies provide insights to previously reported chiral anion phase transfer reactions involving chiral phosphate catalysts in combination with boronic acids.
Keywords: asymmetric catalysis; chiral phosphoric acid; directed reactivity; kinetics; oxa-Michael addition; reaction mechanisms.
Figures
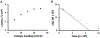
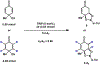
Similar articles
-
Enantioselective synthesis of multisubstituted biaryl skeleton by chiral phosphoric acid catalyzed desymmetrization/kinetic resolution sequence.J Am Chem Soc. 2013 Mar 13;135(10):3964-70. doi: 10.1021/ja311902f. Epub 2013 Feb 26. J Am Chem Soc. 2013. PMID: 23413828
-
Construction of Axially Chiral Compounds via Asymmetric Organocatalysis.Acc Chem Res. 2018 Feb 20;51(2):534-547. doi: 10.1021/acs.accounts.7b00602. Epub 2018 Feb 8. Acc Chem Res. 2018. PMID: 29419282
-
Phase-Transfer and Ion-Pairing Catalysis of Pentanidiums and Bisguanidiniums.Acc Chem Res. 2017 Apr 18;50(4):842-856. doi: 10.1021/acs.accounts.6b00604. Epub 2017 Apr 5. Acc Chem Res. 2017. PMID: 28379012
-
Recent Progress in Asymmetric Relay Catalysis of Metal Complex with Chiral Phosphoric Acid.Top Curr Chem (Cham). 2019 Dec 27;378(1):9. doi: 10.1007/s41061-019-0263-2. Top Curr Chem (Cham). 2019. PMID: 31879793 Review.
-
Recent Advances in Dynamic Kinetic Resolution by Chiral Bifunctional (Thio)urea- and Squaramide-Based Organocatalysts.Molecules. 2016 Oct 14;21(10):1327. doi: 10.3390/molecules21101327. Molecules. 2016. PMID: 27754440 Free PMC article. Review.
Cited by
-
Asymmetric synthesis of CF2-functionalized aziridines by combined strong Brønsted acid catalysis.Beilstein J Org Chem. 2020 Apr 7;16:638-644. doi: 10.3762/bjoc.16.60. eCollection 2020. Beilstein J Org Chem. 2020. PMID: 32318120 Free PMC article.
-
Emergent Organoboron Acid Catalysts.J Org Chem. 2024 Feb 16;89(4):2069-2089. doi: 10.1021/acs.joc.2c01695. Epub 2022 Nov 14. J Org Chem. 2024. PMID: 36374612 Free PMC article. Review.
References
-
- Rauniyar V, Lackner AD, Hamilton GL, Toste FD, Science 2011, 334, 1681–1684; - PubMed
- Shunatona HP, Früh N, Wang Y-M, Rauniyar V, Toste FD, Angew. Chem. Int. Ed 2013, 52, 7724–7727; - PubMed
- Angew. Chem 2013, 125, 7878–7881;
- Lackner AD, Samant AV, Toste FD, J. Am. Chem. Soc 2013, 135, 14090–14093; - PMC - PubMed
- Phipps RJ, Hiramatsu K, Toste FD,J. Am. Chem. Soc 2012, 134, 8376–8379; - PubMed
- Phipps RJ, Toste FD, J. Am. Chem. Soc 2013, 135, 1268– 1271; - PubMed
- Neel AJ, Hehn JP, Tripet PF, Toste FD, J. Am. Chem. Soc 2013, 135, 14044–14047; - PMC - PubMed
- Wang Y-M, Wu J, Hoong C, Rauniyar V, Toste FD, J. Am. Chem. Soc 2012, 134, 12928–12931; - PubMed
- Wu J, Wang Y-M, Drljevic A, Rauniyar V, Phipps RJ, Toste FD, Proc. Natl. Acad. Sci. USA 2013, 110, 13729–13733; - PMC - PubMed
- Yang X, Phipps RJ, Toste FD, J. Am. Chem. Soc 2014, 136, 5225–5228; - PMC - PubMed
- Nelson HM, Reisberg SH, Shunatona HP, Patel JS, Toste FD, Angew. Chem. Int. Ed 2014, 53, 5600–5603; - PMC - PubMed
- Angew. Chem 2014, 126, 5706–5709;
- Nelson HM, Patel JS, Shunatona HP, Toste FD, Chem. Sci 2015, 6, 170–173; - PMC - PubMed
- Nelson HM, Williams BD, Miró J, Toste FD, J. Am. Chem. Soc 2015, 137, 3213–3216; - PMC - PubMed
- Milo A, Neel AJ, Toste FD, Sigman MS, Science 2015, 347, 737–743. - PMC - PubMed
-
-
For other recent reports on CAPT catalysis, see:
- Shen Z, Pan X, Lai Y, Hu J, Wan X, Li X, Zhang H, Xie W, Chem. Sci 2015, 6, 6986–6990; - PMC - PubMed
- Romanov-Michailidis F, Guénée L, Alexakis A, Angew. Chem. Int. Ed 2013, 52, 9266–9270; - PubMed
- Angew. Chem 2013, 125, 9436–9440;
- Xie W, Jiang G, Liu H, Hu J, Pan X,Zhang H, Wan X, Lai Y, Ma D, Angew. Chem. Int. Ed 2013, 52, 12924–12927; - PubMed
- Angew. Chem 2013, 125, 13162–13165;
- Yang Z, He Y, Toste FD, J. Am. Chem. Soc 2016, 138, 9775–9778; - PMC - PubMed
- Yamamoto E,Hilton MJ, Orlandi M, Saini V, Toste FD, Sigman MS, J. Am. Chem. Soc 2016, 138, 15877–15880; - PMC - PubMed
- Avila CM, Patel JS, Reddi Y, Saito M, Nelson HM, Shunatona HP, Sigman MS, Sunoj RB,Toste FD, Angew. Chem. Int. Ed 2017, 56, 5806–5811; - PMC - PubMed
- Angew. Chem 2017, 129, 5900–5905;
- Biswas S, Kubota K, Orlandi M, Turberg M, Miles DH, Sigman MS, Toste FD, Angew. Chem. Int. Ed 2018, 57, 589–593; - PMC - PubMed
- Angew. Chem 2018, 130, 598–602;
- Ye B,Zhao J, Zhao K, McKenna JM, Toste FD, J. Am. Chem. Soc 2018, 140, 8350–8356; - PMC - PubMed
- Coelho JAS, Matsumoto A, Orlandi M, Hilton MJ, Sigman MS,Toste FD, Chem. Sci 2018, 9, 7153–7158. - PMC - PubMed
-
