Obstacles and Coping Strategies of CAR-T Cell Immunotherapy in Solid Tumors
- PMID: 34093592
- PMCID: PMC8170155
- DOI: 10.3389/fimmu.2021.687822
Obstacles and Coping Strategies of CAR-T Cell Immunotherapy in Solid Tumors
Abstract
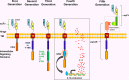
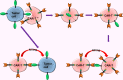
Chimeric antigen receptor (CAR) T-cell immunotherapy refers to an adoptive immunotherapy that has rapidly developed in recent years. It is a novel type of treatment that enables T cells to express specific CARs on their surface, then returns these T cells to tumor patients to kill the corresponding tumor cells. Significant strides in CAR-T cell immunotherapy against hematologic malignancies have elicited research interest among scholars in the treatment of solid tumors. Nonetheless, in contrast with the efficacy of CAR-T cell immunotherapy in the treatment of hematologic malignancies, its general efficacy against solid tumors is insignificant. This has been attributed to the complex biological characteristics of solid tumors. CAR-T cells play a better role in solid tumors, for instance by addressing obstacles including the lack of specific targets, inhibition of tumor microenvironment (TME), homing barriers of CAR-T cells, differentiation and depletion of CAR-T cells, inhibition of immune checkpoints, trogocytosis of CAR-T cells, tumor antigen heterogeneity, etc. This paper reviews the obstacles influencing the efficacy of CAR-T cell immunotherapy in solid tumors, their mechanism, and coping strategies, as well as economic restriction of CAR-T cell immunotherapy and its solutions. It aims to provide some references for researchers to better overcome the obstacles that affect the efficacy of CAR-T cells in solid tumors.
Keywords: chimeric antigen receptor T cell; coping strategies; immunotherapy; obstacles; solid tumors.
Copyright © 2021 Miao, Zhang, Ren, Tang and Li.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures





Similar articles
-
Perspectives on Chimeric Antigen Receptor T-Cell Immunotherapy for Solid Tumors.Front Immunol. 2018 May 22;9:1104. doi: 10.3389/fimmu.2018.01104. eCollection 2018. Front Immunol. 2018. PMID: 29872437 Free PMC article. Review.
-
Genetic Modification of Cytokine Signaling to Enhance Efficacy of CAR T Cell Therapy in Solid Tumors.Front Immunol. 2021 Oct 14;12:738456. doi: 10.3389/fimmu.2021.738456. eCollection 2021. Front Immunol. 2021. PMID: 34721401 Free PMC article. Review.
-
Chimeric antigen receptor-engineered T-cell therapy for liver cancer.Hepatobiliary Pancreat Dis Int. 2018 Aug;17(4):301-309. doi: 10.1016/j.hbpd.2018.05.005. Epub 2018 May 24. Hepatobiliary Pancreat Dis Int. 2018. PMID: 29861325 Review.
-
Immune Cell Hacking: Challenges and Clinical Approaches to Create Smarter Generations of Chimeric Antigen Receptor T Cells.Front Immunol. 2018 Jul 31;9:1717. doi: 10.3389/fimmu.2018.01717. eCollection 2018. Front Immunol. 2018. PMID: 30108584 Free PMC article. Review.
-
CAR T Cells for Solid Tumors: New Strategies for Finding, Infiltrating, and Surviving in the Tumor Microenvironment.Front Immunol. 2019 Feb 5;10:128. doi: 10.3389/fimmu.2019.00128. eCollection 2019. Front Immunol. 2019. PMID: 30804938 Free PMC article. Review.
Cited by
-
Receptor transfer between immune cells by autoantibody-enhanced, CD32-driven trogocytosis is hijacked by HIV-1 to infect resting CD4 T cells.Cell Rep Med. 2024 Apr 16;5(4):101483. doi: 10.1016/j.xcrm.2024.101483. Epub 2024 Apr 4. Cell Rep Med. 2024. PMID: 38579727 Free PMC article.
-
Current status and future challenges of CAR-T cell therapy for osteosarcoma.Front Immunol. 2023 Dec 22;14:1290762. doi: 10.3389/fimmu.2023.1290762. eCollection 2023. Front Immunol. 2023. PMID: 38187386 Free PMC article. Review.
-
Revolutionizing cancer treatment: enhancing CAR-T cell therapy with CRISPR/Cas9 gene editing technology.Front Immunol. 2024 Feb 21;15:1354825. doi: 10.3389/fimmu.2024.1354825. eCollection 2024. Front Immunol. 2024. PMID: 38449862 Free PMC article. Review.
-
Recent findings on chimeric antigen receptor (CAR)-engineered immune cell therapy in solid tumors and hematological malignancies.Stem Cell Res Ther. 2022 Sep 24;13(1):482. doi: 10.1186/s13287-022-03163-w. Stem Cell Res Ther. 2022. PMID: 36153626 Free PMC article. Review.
-
Recent advances and future perspectives of CAR-T cell therapy in head and neck cancer.Front Immunol. 2023 Jun 29;14:1213716. doi: 10.3389/fimmu.2023.1213716. eCollection 2023. Front Immunol. 2023. PMID: 37457699 Free PMC article. Review.
References
-
- Batra SA, Rathi P, Guo L, Courtney AN, Fleurence J, Balzeau J, et al. . Glypican-3-Specific Car T Cells Coexpressing IL15 and IL21 Have Superior Expansion and Antitumor Activity Against Hepatocellular Carcinoma. Cancer Immunol Res (2020) 8(3):309–20. 10.1158/2326-6066.Cir-19-0293 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

