The relative importance of predictive factors for single first-generation EGFR-TKI use for more than 5 years in patients with advanced non-small cell lung cancer: Taiwan multicenter TIPS-5 study
- PMID: 34093743
- PMCID: PMC8142001
- DOI: 10.1177/17588359211018022
The relative importance of predictive factors for single first-generation EGFR-TKI use for more than 5 years in patients with advanced non-small cell lung cancer: Taiwan multicenter TIPS-5 study
Abstract
Background: The relative importance of predictive factors for advanced non-small cell lung cancer (NSCLC) patients on epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitor (TKI) treatment remains unclear.
Materials and methods: We retrospectively enrolled advanced NSCLC patients with single first-generation EGFR-TKI treatment for ⩾5 years (Y) in Taiwan. Clinical data was collected and compared with those of another cohort with single first-line EGFR-TKI treatment for <5 Y. Plasma cell-free DNA (cfDNA) samples were collected from patient subsets, pre- and post-TKI, in the >5 Y group.
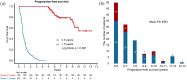
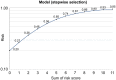
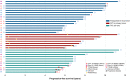
Results: Overall, 128 and 278 patients were enrolled in the ⩾5 Y and <5 Y groups, respectively. Significant factors in the multivariate analysis of patients' characteristics including Eastern Cooperative Oncology Group performance status 0-1, postoperative recurrence, without brain metastasis, oligometastasis (each score of 2), female sex, erlotinib use, and without bone metastasis (each score of 1), were incorporated into a risk scoring system. The area under the receiver operating characteristic curve was 0.82 [95% confidence interval (CI): 0.78-0.86]. Of the plasma cfDNA samples from 33 patients in the ⩾5 Y group, only 1 had a T790M in 25 patients without progressive disease. In 27 patients with single agent use for ⩾96 months, 22 (81.5%) received local treatment (surgery or radiotherapy) for the primary lung tumor before and during TKI treatment.
Conclusion: For NSCLC patients with single first-generation EGFR-TKI use for ⩾5 Y, factors with different relative importance exist and the risk-scoring model is feasible with modest accuracy. The role of local treatment for primary tumors in patients with long-term TKI use requires further investigation.
Keywords: epidermal growth factor receptor; local treatment; more than five years; non-small cell lung cancer; scoring model; tyrosine kinase inhibitor.
© The Author(s), 2021.
Conflict of interest statement
Conflict of interest statement: The authors declare that there is no conflict of interest.
Figures
Similar articles
-
Picoliter-Droplet Digital Polymerase Chain Reaction-Based Analysis of Cell-Free Plasma DNA to Assess EGFR Mutations in Lung Adenocarcinoma That Confer Resistance to Tyrosine-Kinase Inhibitors.Oncologist. 2016 Feb;21(2):156-64. doi: 10.1634/theoncologist.2015-0288. Epub 2016 Jan 14. Oncologist. 2016. PMID: 26768482 Free PMC article.
-
Observational Study of Treatment Patterns in Patients with Epidermal Growth Factor Receptor (EGFR) Mutation-Positive Non-Small Cell Lung Cancer After First-Line EGFR-Tyrosine Kinase Inhibitors.Adv Ther. 2020 Feb;37(2):946-954. doi: 10.1007/s12325-020-01221-4. Epub 2020 Jan 18. Adv Ther. 2020. PMID: 31955357
-
The role of T790M mutation in EGFR-TKI re-challenge for patients with EGFR-mutant advanced lung adenocarcinoma.Oncotarget. 2017 Jan 17;8(3):4994-5002. doi: 10.18632/oncotarget.14007. Oncotarget. 2017. PMID: 27999211 Free PMC article.
-
Neoadjuvant and adjuvant epidermal growth factor receptor tyrosine kinase inhibitor (EGFR-TKI) therapy for lung cancer.Transl Lung Cancer Res. 2015 Feb;4(1):82-93. doi: 10.3978/j.issn.2218-6751.2014.11.08. Transl Lung Cancer Res. 2015. PMID: 25806348 Free PMC article. Review.
-
Gefitinib or Erlotinib vs Chemotherapy for EGFR Mutation-Positive Lung Cancer: Individual Patient Data Meta-Analysis of Overall Survival.J Natl Cancer Inst. 2017 Jun 1;109(6). doi: 10.1093/jnci/djw279. J Natl Cancer Inst. 2017. PMID: 28376144 Review.
Cited by
-
Primary tumor consolidative therapy improves the outcomes of patients with advanced EGFR-mutant lung adenocarcinoma treated with first-line osimertinib.Ther Adv Med Oncol. 2024 Jan 3;16:17588359231220606. doi: 10.1177/17588359231220606. eCollection 2024. Ther Adv Med Oncol. 2024. PMID: 38188463 Free PMC article.
-
Sequential treatment in advanced non-small cell lung cancer harboring EGFR mutations.Ther Adv Respir Dis. 2022 Jan-Dec;16:17534666221132731. doi: 10.1177/17534666221132731. Ther Adv Respir Dis. 2022. PMID: 36305280 Free PMC article.
References
-
- Lynch TJ, Bell DW, Sordella R, et al.. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N Engl J Med 2004; 350: 2129–2139. - PubMed
-
- Paez JG, Janne PA, Lee JC, et al.. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science 2004; 304: 1497–1500. - PubMed
-
- Zhou C, Yao LD. Strategies to improve outcomes of patients with EGRF-mutant non-small cell lung cancer: review of the literature. J Thorac Oncol 2016; 11: 174–186. - PubMed
-
- Park K, Tan EH, O’Byrne K, et al.. Afatinib versus gefitinib as first-line treatment of patients with EGFR mutation-positive non-small-cell lung cancer (LUX-Lung 7): a phase 2B, open-label, randomised controlled trial. Lancet Oncol 2016; 17: 577–589. - PubMed
-
- Gottschling S, Herpel E, Eberhardt WE, et al.. The gefitinib long-term responder (LTR)-a cancer stem-like cell story? Insights from molecular analyses of German long-term responders treated in the IRESSA expanded access program (EAP). Lung Cancer 2012; 77: 183–191. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

