Regulatory control of the Na-Cl co-transporter NCC and its therapeutic potential for hypertension
- PMID: 34094823
- PMCID: PMC8144889
- DOI: 10.1016/j.apsb.2020.09.009
Regulatory control of the Na-Cl co-transporter NCC and its therapeutic potential for hypertension
Abstract
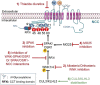
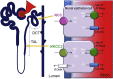
Hypertension is the largest risk factor for cardiovascular disease, the leading cause of mortality worldwide. As blood pressure regulation is influenced by multiple physiological systems, hypertension cannot be attributed to a single identifiable etiology. Three decades of research into Mendelian forms of hypertension implicated alterations in the renal tubular sodium handling, particularly the distal convoluted tubule (DCT)-native, thiazide-sensitive Na-Cl cotransporter (NCC). Altered functions of the NCC have shown to have profound effects on blood pressure regulation as illustrated by the over activation and inactivation of the NCC in Gordon's and Gitelman syndromes respectively. Substantial progress has uncovered multiple factors that affect the expression and activity of the NCC. In particular, NCC activity is controlled by phosphorylation/dephosphorylation, and NCC expression is facilitated by glycosylation and negatively regulated by ubiquitination. Studies have even found parvalbumin to be an unexpected regulator of the NCC. In recent years, there have been considerable advances in our understanding of NCC control mechanisms, particularly via the pathway containing the with-no-lysine [K] (WNK) and its downstream target kinases, SPS/Ste20-related proline-alanine-rich kinase (SPAK) and oxidative stress responsive 1 (OSR1), which has led to the discovery of novel inhibitory molecules. This review summarizes the currently reported regulatory mechanisms of the NCC and discusses their potential as therapeutic targets for treating hypertension.
Keywords: ATP, adenosine triphosphate; Blood pressure regulation; CCC, cation-coupled chloride cotransporters; CCT, conserved carboxy-terminal; CNI, calcineurin inhibitors; CUL3, cullin 3; CUL3/KLHL3-WNK-SPAK/OSR1; Ca2+, calcium ion; Cardiovascular disease; DAG, diacylglycerol; DCT, distal convoluted tubule; DUSP, dual specificity phosphatases; ECF, extracellular fluid; ELISA, enzyme-bound immunosorbent analysis; ERK, extracellular signal-regulated kinases; EnaC, epithelial sodium channels; GABA, gamma-aminobutyric acid; HEK293, human embryonic kidney 293; Hypertension; I1, inhibitor 1; K+, potassium ion; KCC, potassium-chloride-cotransporters; KLHL3, kelch-like 3; KS-WNK1, kidney specific-WNK1; Kinase inhibitors; MAPK, mitogen-activated protein kinase; MO25, mouse protein-25; Membrane trafficking; NCC, sodium–chloride cotransporters; NKCC, sodium–potassium–chloride-cotransporter; Na+, sodium ion; NaCl, sodium chloride; NaCl-cotransporter NCC; OSR1, oxidative stress-responsive gene 1; PCT, proximal convoluted tubule; PHAII, pseudohypoaldosteronism type II; PP, protein phosphatase; PV, parvalbumin; ROMK, renal outer medullary potassium; RasGRP1, RAS guanyl-releasing protein 1; SLC12, solute carrier 12; SPAK, Ste20-related proline-alanine-rich-kinase; TAL, thick ascending limb; Therapeutic targets; WNK, with-no-lysine kinases; mDCT, mammalian DCT; mRNA, messenger RNA.
© 2021 Chinese Pharmaceutical Association and Institute of Materia Medica, Chinese Academy of Medical Sciences. Production and hosting by Elsevier B.V.
Conflict of interest statement
The authors have no conflicts of interest to declare.
Figures


References
-
- Strazzullo P., Galletti F., Barba G. Altered renal handling of sodium in human hypertension: Short review of the evidence. Hypertension. 2003;41:1000–1005. - PubMed
-
- He J., Whelton P.K. Elevated systolic blood pressure and risk of cardiovascular and renal disease: Overview of evidence from observational epidemiologic studies and randomized controlled trials. Am Heart J. 1999;138:211–219. - PubMed
-
- Matsubara M. Renal sodium handling for body fluid maintenance and blood pressure regulation. Yakugaku Zasshi. 2004;124:301–309. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

