Reduction of mechanical loading in tendons induces heterotopic ossification and activation of the β-catenin signaling pathway
- PMID: 34094857
- PMCID: PMC8142054
- DOI: 10.1016/j.jot.2021.03.004
Reduction of mechanical loading in tendons induces heterotopic ossification and activation of the β-catenin signaling pathway
Abstract
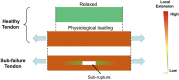
Background: Tendons are the force transferring tissue that enable joint movement. Excessive mechanical loading is commonly considered as a primary factor causing tendinopathy, however, an increasing body of evidence supports the hypothesis that overloading creates microdamage of collagen fibers resulting in a localized decreased loading on the cell population within the damaged site. Heterotopic ossification is a complication of late stage tendinopathy, which can significantly affect the mechanical properties and homeostasis of the tendon. Here, we the examine the effect of mechanical underloading on tendon ossification and investigate its underlying molecular mechanism.
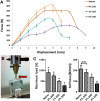
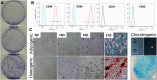
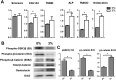
Method: Rabbit Achilles tendons were dissected and cultured in an underloading environment (3% cyclic tensile stain,0.25 Hz, 8 h/day) for either 10, 15 or 20 days. Using isolated tendon-derived stem cells (TDSCs) 3D constructs were generated, cultured and subjected to an underloading environment for 6 days. Histological assessments were performed to evaluate the structure of the 3D constructs; qPCR and immunohistochemistry were employed to study TDSC differentiation and the β-catenin signal pathway was investigated by Western blotting. Mechanical testing was used to determine ability of the tendon to withstand force generation.
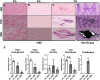
Result: Tendons cultured for extended times in an environment of underloading showed progressive heterotopic ossification and a reduction in biomechanical strength. qPCR revealed that 3D TDSCs constructs cultured in an underloading environment exhibited increased expression of several osteogenic genes: these include RUNX2, ALP and osteocalcin in comparison to tenogenic differentiation markers (scleraxis and tenomodulin). Immunohistochemical analysis further confirmed high osteocalcin production in 3D TDSCs constructs subject to underloading. Western blotting of TDSC constructs revealed that β-catenin accumulation and translocation were associated with an increase in phosphorylation at Ser552 and decrease phosphorylation at Ser33.
Conclusion: These findings unveil a potential mechanism for heterotopic ossification in tendinopathy due to the underloading of TDSCs at the damage sites, and also that β-catenin could be a potential target for treating heterotopic ossification in tendons.
The translational potential: Tendon heterotopic ossification detrimentally affect quality of life especially for those who has atheletic career. This study reveals the possible mechanism of heterotpic ossification in tendon related to mechanical loading. This study provided the possible to develop a mechanical stimulation protocol for preventive and therapeutic purpose for tendon heterotopic ossification.
Keywords: Bioreactor; Heterotopic ossification; Mechanical loading; Mechanobiology; Tendon-derived stem cell.
© 2020 The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


Similar articles
-
3D uniaxial mechanical stimulation induces tenogenic differentiation of tendon-derived stem cells through a PI3K/AKT signaling pathway.FASEB J. 2018 Sep;32(9):4804-4814. doi: 10.1096/fj.201701384R. Epub 2018 Mar 29. FASEB J. 2018. PMID: 29596022
-
Intramuscular injection of Botox causes tendon atrophy by induction of senescence of tendon-derived stem cells.Stem Cell Res Ther. 2021 Jan 7;12(1):38. doi: 10.1186/s13287-020-02084-w. Stem Cell Res Ther. 2021. PMID: 33413592 Free PMC article.
-
Nesfatin-1 Promotes the Osteogenic Differentiation of Tendon-Derived Stem Cells and the Pathogenesis of Heterotopic Ossification in Rat Tendons via the mTOR Pathway.Front Cell Dev Biol. 2020 Dec 3;8:547342. doi: 10.3389/fcell.2020.547342. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 33344440 Free PMC article.
-
Hamstring grafts are tenogenic constructs for ACL reconstruction and Pulsed Electromagnetic Fields improve tendon specific markers expression. An in-vitro study.J Biol Regul Homeost Agents. 2020 Jul-Aug;34(4 Suppl. 3):363-376. Congress of the Italian Orthopaedic Research Society. J Biol Regul Homeost Agents. 2020. PMID: 33261301 Review.
-
Tendon-derived stem cells (TDSCs): from basic science to potential roles in tendon pathology and tissue engineering applications.Stem Cell Rev Rep. 2011 Nov;7(4):883-97. doi: 10.1007/s12015-011-9276-0. Stem Cell Rev Rep. 2011. PMID: 21611803 Review.
Cited by
-
HMGB1 Modulates High Glucose-Induced Erroneous Differentiation of Tendon Stem/Progenitor Cells through RAGE/β-Catenin Pathway.Stem Cells Int. 2024 Apr 9;2024:2335270. doi: 10.1155/2024/2335270. eCollection 2024. Stem Cells Int. 2024. PMID: 38633380 Free PMC article.
-
Heterotopic ossification of the elbow joint in a child: Successful surgical resection - A case report.Heliyon. 2024 Jun 26;10(13):e33756. doi: 10.1016/j.heliyon.2024.e33756. eCollection 2024 Jul 15. Heliyon. 2024. PMID: 39040294 Free PMC article.
-
Erroneous Differentiation of Tendon Stem/Progenitor Cells in the Pathogenesis of Tendinopathy: Current Evidence and Future Perspectives.Stem Cell Rev Rep. 2025 Feb;21(2):423-453. doi: 10.1007/s12015-024-10826-z. Epub 2024 Nov 23. Stem Cell Rev Rep. 2025. PMID: 39579294 Review.
-
Polycystin-1 regulates tendon-derived mesenchymal stem cells fate and matrix organization in heterotopic ossification.Bone Res. 2025 Jan 20;13(1):11. doi: 10.1038/s41413-024-00392-y. Bone Res. 2025. PMID: 39833160 Free PMC article.
-
Mechanically conditioned cell sheets cultured on thermo-responsive surfaces promote bone regeneration.Biomater Transl. 2023 Mar 28;4(1):27-40. doi: 10.12336/biomatertransl.2023.01.005. eCollection 2023. Biomater Transl. 2023. PMID: 37206307 Free PMC article.
References
-
- Sharma P., Maffulli N. Current concepts review tendon injury and tendinopathy: healing and repair. J Bone Jt Surg Am Vol. 2005;87a(1):187–202. - PubMed
-
- Raynor K.J. Ossification of the achilles-tendon. J Am Podiatr Med Assoc. 1986;76(12):688–690. - PubMed
-
- Richards P.J. Achilles tendon ossification: pathology, imaging and aetiology. Disabil Rehabil. 2008;30(20–22):1651–1665. - PubMed
-
- Xu R.S. Heterotopic ossification: mechanistic insights and clinical challenges. Bone. 2018;109:134–142. - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

