lncRNA MIAT/HMGB1 Axis Is Involved in Cisplatin Resistance via Regulating IL6-Mediated Activation of the JAK2/STAT3 Pathway in Nasopharyngeal Carcinoma
- PMID: 34094941
- PMCID: PMC8173225
- DOI: 10.3389/fonc.2021.651693
lncRNA MIAT/HMGB1 Axis Is Involved in Cisplatin Resistance via Regulating IL6-Mediated Activation of the JAK2/STAT3 Pathway in Nasopharyngeal Carcinoma
Abstract
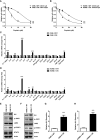
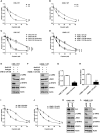
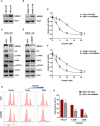
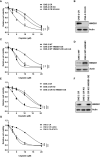
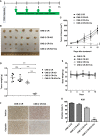
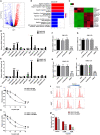
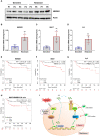
Cisplatin-based chemotherapy and radiotherapy are the main first-line treatment strategies for nasopharyngeal carcinoma (NPC) patients. Unfortunately, resistance is a major obstacle in the clinical management of NPC patients. We prove that the expression level of high-mobility group box 1 (HMGB1) is dramatically increased in resistant NPC cells than that in sensitive cells. HMGB1 induces the expression and secretion of IL6, which leads to constitutive autocrine activation of the JAK2/STAT3 pathway and eventually contributes to chemoresistance in NPC cells. Long non-coding RNAs (lncRNAs) have been identified as key regulators involved in drug resistance. In this study, using GO analysis of the biological process and differential expression analysis, we find 12 significantly altered IncRNAs in NPC cell lines, which may be involved in regulating gene expression. Furthermore, we determine that elevated lncRNA MIAT level upregulates HMGB1 expression, contributing to cisplatin resistance in NPC cells. We find that the deficiency of the lncRNA MIAT/HMGB1 axis, inhibition of JAK2/STAT3, or neutralization of IL6 by antibodies significantly re-sensitizes resistant NPC cells to cisplatin in resistant NPC cells. Moreover, we provide the in vivo evidence that the deficiency of HMGB1 reduces cisplatin-resistant tumor growth. Most importantly, we provide clinical evidence showing that the expression level of the lncRNA MIAT/HMGB1/IL6 axis is elevated in resistant NPC tumors, which is highly correlated with poor clinical outcome. Our findings identify a novel chemoresistance mechanism regulated by the lncRNA MIAT/HMGB1/IL6 axis, which indicates the possibilities for lncRNA MIAT, HMGB1, and IL6 as biomarkers for chemoresistance and targets for developing novel strategies to overcome resistance in NPC patients.
Keywords: HMGB1; IL6; NPC; cisplatin resistance; lncRNA MIAT.
Copyright © 2021 Zhu, Liu, Wang, Cong, Lin, Wang, Liu, Wang, Yang and Li.
Conflict of interest statement
Author YSW was employed by the company Harbin Boshixuan Technology Co. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest
Figures
Similar articles
-
Inhibition of HMGB1 Overcomes Resistance to Radiation and Chemotherapy in Nasopharyngeal Carcinoma.Onco Targets Ther. 2020 May 14;13:4189-4199. doi: 10.2147/OTT.S239243. eCollection 2020. Onco Targets Ther. 2020. PMID: 32523355 Free PMC article.
-
Next generation deep sequencing identified a novel lncRNA n375709 associated with paclitaxel resistance in nasopharyngeal carcinoma.Oncol Rep. 2016 Oct;36(4):1861-7. doi: 10.3892/or.2016.4981. Epub 2016 Jul 28. Oncol Rep. 2016. PMID: 27498905 Free PMC article.
-
LncRNA MIAT facilitates osteosarcoma progression by regulating mir-128-3p/VEGFC axis.IUBMB Life. 2019 Jul;71(7):845-853. doi: 10.1002/iub.2001. Epub 2019 Jan 10. IUBMB Life. 2019. PMID: 30629798
-
LncRNA MIAT correlates with immune infiltrates and drug reactions in hepatocellular carcinoma.Int Immunopharmacol. 2020 Dec;89(Pt A):107071. doi: 10.1016/j.intimp.2020.107071. Epub 2020 Nov 19. Int Immunopharmacol. 2020. PMID: 33221703
-
Role of long non-coding RNA in chemoradiotherapy resistance of nasopharyngeal carcinoma.Front Oncol. 2024 Feb 29;14:1346413. doi: 10.3389/fonc.2024.1346413. eCollection 2024. Front Oncol. 2024. PMID: 38487724 Free PMC article. Review.
Cited by
-
CircMMP11 as a prognostic biomarker mediates miR-361-3p/HMGB1 axis to accelerate malignant progression of hepatocellular carcinoma.Open Med (Wars). 2023 Nov 2;18(1):20230803. doi: 10.1515/med-2023-0803. eCollection 2023. Open Med (Wars). 2023. PMID: 38025527 Free PMC article.
-
Insulin/IGF Axis and the Receptor for Advanced Glycation End Products: Role in Meta-inflammation and Potential in Cancer Therapy.Endocr Rev. 2023 Jul 11;44(4):693-723. doi: 10.1210/endrev/bnad005. Endocr Rev. 2023. PMID: 36869790 Free PMC article. Review.
-
Multiple functions of HMGB1 in cancer.Front Oncol. 2024 Apr 25;14:1384109. doi: 10.3389/fonc.2024.1384109. eCollection 2024. Front Oncol. 2024. PMID: 38725632 Free PMC article. Review.
-
Potential prognostic value of a eight ferroptosis-related lncRNAs model and the correlative immune activity in oral squamous cell carcinoma.BMC Genom Data. 2022 Nov 16;23(1):80. doi: 10.1186/s12863-022-01097-z. BMC Genom Data. 2022. PMID: 36384476 Free PMC article.
-
Involvement of Non-Coding RNAs in Chemo- and Radioresistance of Nasopharyngeal Carcinoma.Cancer Manag Res. 2021 Nov 23;13:8781-8794. doi: 10.2147/CMAR.S336265. eCollection 2021. Cancer Manag Res. 2021. PMID: 34849030 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

