Conversion from cilostazol to OPC-13015 linked to mitigation of cognitive impairment
- PMID: 34095441
- PMCID: PMC8158162
- DOI: 10.1002/trc2.12182
Conversion from cilostazol to OPC-13015 linked to mitigation of cognitive impairment
Abstract
Introduction: Cilostazol may be a novel therapeutic agent for Alzheimer's disease. Its metabolite, OPC-13015, has a stronger inhibitory effect on type 3 phosphodiesterase than cilostazol.
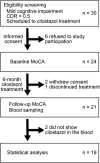
Methods: We prospectively enrolled patients with mild cognitive impairment to whom cilostazol was newly prescribed. Patients underwent the Montreal Cognitive Assessment (MoCA) twice, at a 6-month interval. Plasma cilostazol, OPC-13015, OPC-13213, and OPC-13217 concentrations were determined using liquid chromatography-tandem mass spectrometry.
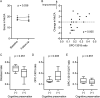
Results: MoCA score changes from baseline to the 6-month visit were positively correlated with ratios of OPC-13015 to cilostazol and total metabolites (n = 19, P = .005). Patients with higher ratios of OPC-13015 (≥0.18, median value; n = 10) had significantly higher MoCA scores (P = .036) than patients with lower ratios (the ratio <0.18, n = 9). The absolute value of OPC-13015 concentration in blood was also higher in patients with preserved cognitive function (P = .033).
Discussion: Blood OPC-13015 levels may be a predictive biomarker of cilostazol treatment for Alzheimer's disease.
Keywords: Alzheimer's disease; OPC‐13015; cilostazol; drug repositioning; mild cognitive impairment; stratified medicine.
© 2021 The Authors. Alzheimer's & Dementia: Translational Research & Clinical Interventions published by Wiley Periodicals, Inc. on behalf of Alzheimer's Association.
Conflict of interest statement
Mr. Kawakami and Mr. Yamamoto are employees of Shimadzu Corporation, an analytical instrument manufacturer that is not involved in the sale of cilostazol. Dr. Ihara has a patent concerning cilostazol (WO2013187075A1) titled “Prophylactic and/or therapeutic agent for mild cognitive impairment.” Dr. Saito, Mr. Kawakami, Mr. Yamamoto, and Dr. Ihara have applied for a patent concerning the usefulness of evaluating the levels of cilostazol and its metabolites. In addition, Dr. Ihara reports grants from Panasonic, BMS, and Otsuka Pharmaceutical, and personal fees from Daiichi Sankyo Co Ltd, Eisai, and Bayer, which were not involved in the current work. The other authors declare no competing financial interests.
Figures


Similar articles
-
Impact of Cilostazol Pharmacokinetics on the Development of Cardiovascular Side Effects in Patients with Cerebral Infarction.Biol Pharm Bull. 2021;44(11):1767-1774. doi: 10.1248/bpb.b21-00535. Biol Pharm Bull. 2021. PMID: 34719653
-
Simultaneous quantitative determination of cilostazol and its metabolites in human plasma by high-performance liquid chromatography.J Chromatogr B Biomed Sci Appl. 1999 May 28;728(2):251-62. doi: 10.1016/s0378-4347(99)00104-8. J Chromatogr B Biomed Sci Appl. 1999. PMID: 10406210
-
A multicenter, randomized, placebo-controlled trial for cilostazol in patients with mild cognitive impairment: The COMCID study protocol.Alzheimers Dement (N Y). 2016 Oct 27;2(4):250-257. doi: 10.1016/j.trci.2016.10.001. eCollection 2016 Nov. Alzheimers Dement (N Y). 2016. PMID: 29067312 Free PMC article.
-
The metabolism of a new antithrombotic and vasodilating agent, cilostazol, in rat, dog and man.Arzneimittelforschung. 1985;35(7A):1133-40. Arzneimittelforschung. 1985. PMID: 4074424
-
PDE3 Inhibitors Repurposed as Treatments for Age-Related Cognitive Impairment.Mol Neurobiol. 2019 Jun;56(6):4306-4316. doi: 10.1007/s12035-018-1374-4. Epub 2018 Oct 11. Mol Neurobiol. 2019. PMID: 30311144 Review.
Cited by
-
Effects of cilostazol on cognitive function and dementia risk: A systematic review and meta-analysis.Medicine (Baltimore). 2024 Dec 13;103(50):e40668. doi: 10.1097/MD.0000000000040668. Medicine (Baltimore). 2024. PMID: 39686486 Free PMC article.
-
Relationship Between RNF213 p.R4810K and Echocardiographic Findings in Patients with Cerebrovascular Diseases: A Multicenter Prospective Cohort Study.J Am Heart Assoc. 2025 Feb 4;14(3):e036333. doi: 10.1161/JAHA.124.036333. Epub 2025 Jan 27. J Am Heart Assoc. 2025. PMID: 39868517 Free PMC article.
-
Efficacy and Safety of Cilostazol in Mild Cognitive Impairment: A Randomized Clinical Trial.JAMA Netw Open. 2023 Dec 1;6(12):e2344938. doi: 10.1001/jamanetworkopen.2023.44938. JAMA Netw Open. 2023. PMID: 38048134 Free PMC article. Clinical Trial.
-
RNF213 p.R4810K Variant Carriers with Intracranial Arterial Stenosis Have a Low Atherosclerotic Burden.J Atheroscler Thromb. 2022 Nov 1;29(11):1655-1662. doi: 10.5551/jat.63379. Epub 2021 Dec 25. J Atheroscler Thromb. 2022. PMID: 34955493 Free PMC article.
References
LinkOut - more resources
Full Text Sources
