β-sitosterol reduces anxiety and synergizes with established anxiolytic drugs in mice
- PMID: 34095883
- PMCID: PMC8149471
- DOI: 10.1016/j.xcrm.2021.100281
β-sitosterol reduces anxiety and synergizes with established anxiolytic drugs in mice
Abstract
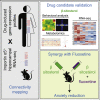
Anxiety and stress-related conditions represent a significant health burden in modern society. Unfortunately, most anxiolytic drugs are prone to side effects, limiting their long-term usage. Here, we employ a bioinformatics screen to identify drugs for repurposing as anxiolytics. Comparison of drug-induced gene-expression profiles with the hippocampal transcriptome of an importin α5 mutant mouse model with reduced anxiety identifies the hypocholesterolemic agent β-sitosterol as a promising candidate. β-sitosterol activity is validated by both intraperitoneal and oral application in mice, revealing it as the only clear anxiolytic from five closely related phytosterols. β-sitosterol injection reduces the effects of restraint stress, contextual fear memory, and c-Fos activation in the prefrontal cortex and dentate gyrus. Moreover, synergistic anxiolysis is observed when combining sub-efficacious doses of β-sitosterol with the SSRI fluoxetine. These preclinical findings support further development of β-sitosterol, either as a standalone anxiolytic or in combination with low-dose SSRIs.
Keywords: CNS drugs; SSRIs; anxiolytics; drug repositioning; fluoxetine; metabolomics; phytosterol; transcriptomics; β-Sitosterol.
© 2021 The Author(s).
Conflict of interest statement
N.P. and M.F. have a patent application related to this work, PCT patent application number PCT/IL2018/050495, Publication number WO/2018/207178, on “Methods of treating psychiatric stress disorders.”
Figures




Similar articles
-
Effect of Early-Life Fluoxetine on Anxiety-Like Behaviors in BDNF Val66Met Mice.Am J Psychiatry. 2017 Dec 1;174(12):1203-1213. doi: 10.1176/appi.ajp.2017.15121592. Epub 2017 Oct 31. Am J Psychiatry. 2017. PMID: 29084453 Free PMC article.
-
Influence of anxiolytic drugs on the effects of specific serotonin reuptake inhibitors in the forced swimming test in mice.J Psychopharmacol. 1997;11(3):211-8. doi: 10.1177/026988119701100303. J Psychopharmacol. 1997. PMID: 9305412
-
Target brain sites of the anxiolytic effect of citalopram, a selective serotonin reuptake inhibitor.Eur J Pharmacol. 2006 Mar 18;534(1-3):129-32. doi: 10.1016/j.ejphar.2005.12.073. Epub 2006 Feb 21. Eur J Pharmacol. 2006. PMID: 16494863
-
[Biological basis of anxiety disorders and serotonergic anxiolytics].Nihon Shinkei Seishin Yakurigaku Zasshi. 2004 Jun;24(3):125-31. Nihon Shinkei Seishin Yakurigaku Zasshi. 2004. PMID: 15291241 Review. Japanese.
-
Can the antidysphoric and anxiolytic profiles of selective serotonin reuptake inhibitors be related to their ability to increase brain 3 alpha, 5 alpha-tetrahydroprogesterone (allopregnanolone) availability?Biol Psychiatry. 1998 Nov 1;44(9):865-73. doi: 10.1016/s0006-3223(98)00070-5. Biol Psychiatry. 1998. PMID: 9807641 Review.
Cited by
-
A Causal Inference Study of Circulating Metabolites Mediating the Effect of Obesity-Related Indicators on the Incidence of Anxiety Disorders.Brain Behav. 2025 Jul;15(7):e70653. doi: 10.1002/brb3.70653. Brain Behav. 2025. PMID: 40619960 Free PMC article.
-
β-sitosterol alleviates dextran sulfate sodium-induced experimental colitis via inhibition of NLRP3/Caspase-1/GSDMD-mediated pyroptosis.Front Pharmacol. 2023 Oct 26;14:1218477. doi: 10.3389/fphar.2023.1218477. eCollection 2023. Front Pharmacol. 2023. PMID: 37954856 Free PMC article.
-
Liver Extracellular Vesicles and Particles Enriched β-Sitosterol Effectively Promote Liver Regeneration in Mice.Int J Nanomedicine. 2024 Aug 8;19:8117-8137. doi: 10.2147/IJN.S465346. eCollection 2024. Int J Nanomedicine. 2024. PMID: 39139504 Free PMC article.
-
Network pharmacology, molecular docking, and experimental validation to explore the potential mechanism of Long Mu Qing Xin mixture for the treatment of attention deficit hyperactivity disorder.Front Pharmacol. 2023 Mar 17;14:1144907. doi: 10.3389/fphar.2023.1144907. eCollection 2023. Front Pharmacol. 2023. PMID: 37007045 Free PMC article.
-
Network Pharmacology, Molecular Docking, and Experimental Validation on Guiluoshi Anzang Decoction Against Premature Ovarian Insufficiency.Comb Chem High Throughput Screen. 2025;28(4):724-736. doi: 10.2174/0113862073291139240506114446. Comb Chem High Throughput Screen. 2025. PMID: 38757315
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

