Molecular basis for recognition of the Group A Carbohydrate backbone by the PlyC streptococcal bacteriophage endolysin
- PMID: 34096588
- PMCID: PMC8555655
- DOI: 10.1042/BCJ20210158
Molecular basis for recognition of the Group A Carbohydrate backbone by the PlyC streptococcal bacteriophage endolysin
Abstract
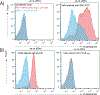
Endolysins are peptidoglycan (PG) hydrolases that function as part of the bacteriophage (phage) lytic system to release progeny phage at the end of a replication cycle. Notably, endolysins alone can produce lysis without phage infection, which offers an attractive alternative to traditional antibiotics. Endolysins from phage that infect Gram-positive bacterial hosts contain at least one enzymatically active domain (EAD) responsible for hydrolysis of PG bonds and a cell wall binding domain (CBD) that binds a cell wall epitope, such as a surface carbohydrate, providing some degree of specificity for the endolysin. Whilst the EADs typically cluster into conserved mechanistic classes with well-defined active sites, relatively little is known about the nature of the CBDs and only a few binding epitopes for CBDs have been elucidated. The major cell wall components of many streptococci are the polysaccharides that contain the polyrhamnose (pRha) backbone modified with species-specific and serotype-specific glycosyl side chains. In this report, using molecular genetics, microscopy, flow cytometry and lytic activity assays, we demonstrate the interaction of PlyCB, the CBD subunit of the streptococcal PlyC endolysin, with the pRha backbone of the cell wall polysaccharides, Group A Carbohydrate (GAC) and serotype c-specific carbohydrate (SCC) expressed by the Group A Streptococcus and Streptococcus mutans, respectively.
Keywords: Streptococcus pyogenes; bacteriophage; cell wall; endolysin; polysaccharide; rhamnose.
© 2021 The Author(s). Published by Portland Press Limited on behalf of the Biochemical Society.
Figures



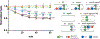
Similar articles
-
High avidity drives the interaction between the streptococcal C1 phage endolysin, PlyC, with the cell surface carbohydrates of Group A Streptococcus.Mol Microbiol. 2021 Aug;116(2):397-415. doi: 10.1111/mmi.14719. Epub 2021 Apr 3. Mol Microbiol. 2021. PMID: 33756056
-
Potential Role of the Host-Derived Cell-Wall Binding Domain of Endolysin CD16/50L as a Molecular Anchor in Preservation of Uninfected Clostridioides difficile for New Rounds of Phage Infection.Microbiol Spectr. 2022 Apr 27;10(2):e0236121. doi: 10.1128/spectrum.02361-21. Epub 2022 Apr 4. Microbiol Spectr. 2022. PMID: 35377223 Free PMC article.
-
Structural insights into the binding and catalytic mechanisms of the Listeria monocytogenes bacteriophage glycosyl hydrolase PlyP40.Mol Microbiol. 2018 Apr;108(2):128-142. doi: 10.1111/mmi.13922. Epub 2018 Mar 6. Mol Microbiol. 2018. PMID: 29405497
-
Phage Lysis: Multiple Genes for Multiple Barriers.Adv Virus Res. 2019;103:33-70. doi: 10.1016/bs.aivir.2018.09.003. Epub 2018 Nov 28. Adv Virus Res. 2019. PMID: 30635077 Free PMC article. Review.
-
The pneumococcal cell wall degrading enzymes: a modular design to create new lysins?Microb Drug Resist. 1997 Summer;3(2):199-211. doi: 10.1089/mdr.1997.3.199. Microb Drug Resist. 1997. PMID: 9185148 Review.
Cited by
-
The role of nanocomposites against biofilm infections in humans.Front Cell Infect Microbiol. 2023 Feb 28;13:1104615. doi: 10.3389/fcimb.2023.1104615. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 36926513 Free PMC article. Review.
-
Treating Bacterial Infections with Bacteriophage-Based Enzybiotics: In Vitro, In Vivo and Clinical Application.Antibiotics (Basel). 2021 Dec 6;10(12):1497. doi: 10.3390/antibiotics10121497. Antibiotics (Basel). 2021. PMID: 34943709 Free PMC article. Review.
-
Direct Lytic Agents: Novel, Rapidly Acting Potential Antimicrobial Treatment Modalities for Systemic Use in the Era of Rising Antibiotic Resistance.Front Microbiol. 2022 Mar 3;13:841905. doi: 10.3389/fmicb.2022.841905. eCollection 2022. Front Microbiol. 2022. PMID: 35308352 Free PMC article. Review.
-
Structure and mechanism of biosynthesis of Streptococcus mutans cell wall polysaccharide.Nat Commun. 2025 Jan 22;16(1):954. doi: 10.1038/s41467-025-56205-1. Nat Commun. 2025. PMID: 39843487 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

