Sequential activation of uterine epithelial IGF1R by stromal IGF1 and embryonic IGF2 directs normal uterine preparation for embryo implantation
- PMID: 34097060
- PMCID: PMC8648386
- DOI: 10.1093/jmcb/mjab034
Sequential activation of uterine epithelial IGF1R by stromal IGF1 and embryonic IGF2 directs normal uterine preparation for embryo implantation
Abstract
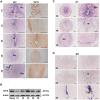
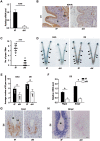
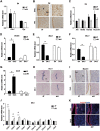
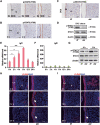
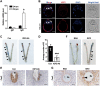
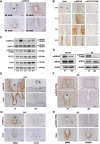
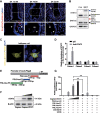
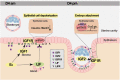
Embryo implantation in both humans and rodents is initiated by the attachment of a blastocyst to the uterine epithelium. For blastocyst attachment, the uterine epithelium needs to transform at both the structural and molecular levels first, and then initiate the interaction with trophectoderm. Any perturbation during this process will result in implantation failure or long-term adverse pregnancy outcomes. Endocrine steroid hormones, which function through nuclear receptors, combine with the local molecules produced by the uteri or embryo to facilitate implantation. The insulin-like growth factor (IGF) signaling has been reported to play a vital role during pregnancy. However, its physiological function during implantation remains elusive. This study revealed that mice with conditional deletion of Igf1r gene in uteri suffered from subfertility, mainly due to the disturbed uterine receptivity and abnormal embryo implantation. Mechanistically, we uncovered that in response to the nidatory estrogen on D4 of pregnancy, the epithelial IGF1R, stimulated by the stromal cell-produced IGF1, facilitated epithelial STAT3 activation to modulate the epithelial depolarity. Furthermore, embryonic derived IGF2 could activate both the epithelial ERK1/2 and STAT3 signaling through IGF1R, which was critical for the transcription of Cox2 and normal attachment reaction. In brief, our data revealed that epithelial IGF1R was sequentially activated by the uterine stromal IGF1 and embryonic IGF2 to guarantee normal epithelium differentiation during the implantation process.
Keywords: IGF1; IGF1R; IGF2; blastocyst; implantation; uterine epithelium.
© The Author(s) (2021). Published by Oxford University Press on behalf of Journal of Molecular Cell Biology, CEMCS, CAS.
Figures
References
-
- Auernhammer C.J., Melmed S. (2000). Leukemia-inhibitory factor—neuroimmune modulator of endocrine function. Endocr. Rev. 21, 313–345. - PubMed
-
- Chen J.R., Cheng J.G., Shatzer T., et al. (2000). Leukemia inhibitory factor can substitute for nidatory estrogen and is essential to inducing a receptive uterus for implantation but is not essential for subsequent embryogenesis. Endocrinology 141, 4365–4372. - PubMed
-
- Constância M., Hemberger M., Hughes J., et al. (2002). Placental-specific IGF-II is a major modulator of placental and fetal growth. Nature 417, 945–948. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

