Chemodivergent manganese-catalyzed C-H activation: modular synthesis of fluorogenic probes
- PMID: 34099672
- PMCID: PMC8185085
- DOI: 10.1038/s41467-021-23462-9
Chemodivergent manganese-catalyzed C-H activation: modular synthesis of fluorogenic probes
Abstract
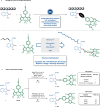
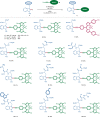
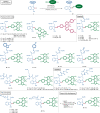
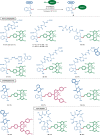
Bioorthogonal late-stage diversification of amino acids and peptides bears enormous potential for drug discovery and molecular imaging. Despite major accomplishments, these strategies largely rely on traditional, lengthy prefunctionalization methods, heavily involving precious transition-metal catalysis. Herein, we report on a resource-economical manganese(I)-catalyzed C-H fluorescent labeling of structurally complex peptides ensured by direct alkynylation and alkenylation manifolds. This modular strategy sets the stage for unraveling structure-activity relationships between structurally discrete fluorophores towards the rational design of BODIPY fluorogenic probes for real-time analysis of immune cell function.
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Fluorescent coumarin-alkynes for labeling of amino acids and peptides via manganese(I)-catalyzed C-H alkenylation.Chem Commun (Camb). 2024 May 16;60(41):5423-5426. doi: 10.1039/d4cc00361f. Chem Commun (Camb). 2024. PMID: 38683668
-
Late-stage stitching enabled by manganese-catalyzed C─H activation: Peptide ligation and access to cyclopeptides.Sci Adv. 2021 Feb 26;7(9):eabe6202. doi: 10.1126/sciadv.abe6202. Print 2021 Feb. Sci Adv. 2021. PMID: 33637533 Free PMC article.
-
Late-Stage Peptide Diversification by Position-Selective C-H Activation.Angew Chem Int Ed Engl. 2018 Nov 5;57(45):14700-14717. doi: 10.1002/anie.201806250. Epub 2018 Oct 9. Angew Chem Int Ed Engl. 2018. PMID: 29969532 Review.
-
Bioorthogonal Diversification of Peptides through Selective Ruthenium(II)-Catalyzed C-H Activation.Angew Chem Int Ed Engl. 2017 Feb 1;56(6):1576-1580. doi: 10.1002/anie.201609631. Epub 2017 Jan 11. Angew Chem Int Ed Engl. 2017. PMID: 28074503
-
The emergence of the C-H functionalization strategy in medicinal chemistry and drug discovery.Chem Commun (Camb). 2021 Oct 19;57(83):10842-10866. doi: 10.1039/d1cc04083a. Chem Commun (Camb). 2021. PMID: 34596175 Review.
Cited by
-
Homogeneous Manganese-Catalyzed Hydrofunctionalizations of Alkenes and Alkynes: Catalytic and Mechanistic Tendencies.ACS Omega. 2022 Oct 11;7(42):37008-37038. doi: 10.1021/acsomega.2c05109. eCollection 2022 Oct 25. ACS Omega. 2022. PMID: 36312376 Free PMC article. Review.
-
Clickable tryptophan modification for late-stage diversification of native peptides.Sci Adv. 2024 Jul 12;10(28):eadp9958. doi: 10.1126/sciadv.adp9958. Epub 2024 Jul 10. Sci Adv. 2024. PMID: 38985871 Free PMC article.
-
A Bivalent Activatable Fluorescent Probe for Screening and Intravital Imaging of Chemotherapy-Induced Cancer Cell Death.Angew Chem Weinheim Bergstr Ger. 2022 Jan 26;134(5):e202113020. doi: 10.1002/ange.202113020. Epub 2021 Dec 16. Angew Chem Weinheim Bergstr Ger. 2022. PMID: 38505298 Free PMC article.
-
Transition-Metal-Catalyzed C-H Bond Activation for the Formation of C-C Bonds in Complex Molecules.Chem Rev. 2023 Jun 28;123(12):7692-7760. doi: 10.1021/acs.chemrev.2c00888. Epub 2023 May 10. Chem Rev. 2023. PMID: 37163671 Free PMC article. Review.
-
Modular synthesis of clickable peptides via late-stage maleimidation on C(7)-H tryptophan.Nat Commun. 2023 Jul 5;14(1):3973. doi: 10.1038/s41467-023-39703-y. Nat Commun. 2023. PMID: 37407547 Free PMC article.
References
-
- Kuter K, et al. Adaptation within mitochondrial oxidative phosphorylation supercomplexes and membrane viscosity during degeneration of dopaminergic neurons in an animal model of early Parkinson’s disease. Biochim. Biophys. Acta, Mol. Basis Dis. 2016;1862:741–753. doi: 10.1016/j.bbadis.2016.01.022. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

