Hydroxychloroquine plus standard of care compared with standard of care alone in COVID-19: a meta-analysis of randomized controlled trials
- PMID: 34099745
- PMCID: PMC8184930
- DOI: 10.1038/s41598-021-91089-3
Hydroxychloroquine plus standard of care compared with standard of care alone in COVID-19: a meta-analysis of randomized controlled trials
Abstract
The efficacy and safety of Hydroxychloroquine (HCQ) in treating coronavirus disease (COVID-19) is disputed. This systematic review and meta-analysis aimed to examine the efficacy and safety of HCQ in addition to standard of care (SOC) in COVID-19. PubMed, the Cochrane Library, Embase, Web of sciences, and medRxiv were searched up to March 15, 2021. Clinical studies registry databases were also searched for identifying potential clinical trials. The references list of the key studies was reviewed to identify additional relevant resources. The quality of the included studies was evaluated using the Cochrane Collaboration tool and Jadad checklist. Meta-analysis was performed using RevMan software (version 5.3). Eleven randomized controlled trials with a total number of 8161 patients were identified as eligible for meta-analysis. No significant differences were observed between the two treatment groups in terms of negative rate of polymerase chain reaction (PCR) (Risk ratio [RR]: 0.99, 95% confidence interval (CI) 0.90, 1.08; P = 0.76), PCR negative conversion time (Mean difference [MD]: - 1.06, 95% CI - 3.10, 0.97; P = 0.30), all-cause mortality (RR: 1.09, 95% CI 1.00, 1.20; P = 0.06), body temperature recovery time (MD: - 0.64, 95% CI - 1.37, 0.10; P = 0.09), length of hospital stay (MD: - 0.17, 95% CI - 0.80, 0.46; P = 0.59), use of mechanical ventilation (RR: 1.12, 95% CI 0.95, 1.32; P = 0.19), and disease progression (RR = 0.82, 95% CI 0.37, 1.85; P = 0.64). However, there was a significant difference between two groups regarding adverse events (RR: 1.81, 95% CI 1.36, 2.42; P < 0.05). The findings suggest that the addition of HCQ to SOC has no benefit in the treatment of hospitalized patients with COVID-19. Additionally, it is associated with more adverse events.
Conflict of interest statement
The authors declare no competing interests.
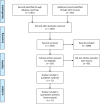
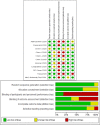
Figures


References
-
- Rao, K. et al. Review on newly identified coronavirus and its genomic organization. (2020).
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

