Improvement in extracellular secretion of recombinant L-asparaginase II by Escherichia coli BL21 (DE3) using glycine and n-dodecane
- PMID: 34100260
- PMCID: PMC8324600
- DOI: 10.1007/s42770-021-00534-y
Improvement in extracellular secretion of recombinant L-asparaginase II by Escherichia coli BL21 (DE3) using glycine and n-dodecane
Abstract
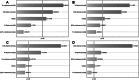
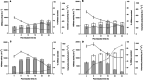
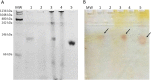
L-asparaginase II (ASNase) is the biopharmaceutical of choice for the treatment of acute lymphoblastic leukaemia. In this study, E. coli BL21 (DE3) transformed with the pET15b + asnB vector which expresses recombinant ASNase was used as a source to obtain this enzyme. The ideal conditions to produce ASNase would be a high level of secretion into the extracellular medium, which depends not only on the application of molecular biology techniques but also on the development of a strategy to modify cell permeability such as the addition of substances to the culture medium that stimulate destabilisation of structural components of the cell. Thus, the growth of E. coli BL21 (DE3) in modified Luria-Bertani broth, supplemented with 0.8% (w/v) glycine and 6% (v/v) n-dodecane, increased the total yield of ASNase by about 50% (15,108 IU L-1) and resulted in a 16-fold increase in extracellular enzymatic productivity (484 IU L-1 h-1), compared to production using the same medium without addition of these substances. Most of the enzyme (89%) was secreted into the culture medium 24 h after the induction step. This proposed approach presents a simple strategy to increase extracellular production of ASNase in E. coli.
Keywords: Culture medium; Dodecane; Glycine; Luria–Bertani; Permeabilisation.
© 2021. Sociedade Brasileira de Microbiologia.
Figures



References
-
- Egler R, Ahuja S, Matloub Y (2016) L-asparaginase in the treatment of patients with acute lymphoblastic leukemia. J Pharmacol Pharmacother [Internet] 7(2):62. Available from: http://www.jpharmacol.com/text.asp?2016/7/2/62/184769 . Accessed 05 June 2021 - PMC - PubMed
-
- Bae N, Pollak A, Lubec G (2011) Proteins from Erwinia asparaginase Erwinase ® and E. coli asparaginase 2 MEDAC ® for treatment of human leukemia, show a multitude of modifications for which the consequences are completely unclear. Electrophoresis [Internet] [cited 2014 May 22];32(14):1824–8. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21769889. Accessed 05 June 2021 - PubMed
-
- Pieters R, Appel I, Kuehnel H-J, Tetzlaff-Fohr I, Pichlmeier U, van der Vaart I, et al. () Pharmacokinetics, pharmacodynamics, efficacy, and safety of a new recombinant asparaginase preparation in children with previously untreated acute lymphoblastic leukemia: a randomized phase 2 clinical trial. Blood [Internet] [cited 2014 May 7];112(13):4832–8. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18805963. Accessed 05 June 2021 - PubMed
-
- Chessells J, Saha V. Childhood lymphoblastic leukemia. In: Pinkerton R, Shankar A, Matthay K, editors. Evidence-based Pediatric Oncology. 2. Malden: Blackwell; 2007. pp. 267–278.
-
- Lundbeck, Deerfield, IL U. Discontinnuation of Elspar [Internet]. 2012 [cited 2016 Jul 1]. p. 1. Available from: http://www.fda.gov/downloads/Drugs/DrugSafety/DrugShortages/UCM321556.pdf. Accessed 05 June 2021
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

