Bladder cancer therapy using a conformationally fluid tumoricidal peptide complex
- PMID: 34103518
- PMCID: PMC8187399
- DOI: 10.1038/s41467-021-23748-y
Bladder cancer therapy using a conformationally fluid tumoricidal peptide complex
Abstract
Partially unfolded alpha-lactalbumin forms the oleic acid complex HAMLET, with potent tumoricidal activity. Here we define a peptide-based molecular approach for targeting and killing tumor cells, and evidence of its clinical potential (ClinicalTrials.gov NCT03560479). A 39-residue alpha-helical peptide from alpha-lactalbumin is shown to gain lethality for tumor cells by forming oleic acid complexes (alpha1-oleate). Nuclear magnetic resonance measurements and computational simulations reveal a lipid core surrounded by conformationally fluid, alpha-helical peptide motifs. In a single center, placebo controlled, double blinded Phase I/II interventional clinical trial of non-muscle invasive bladder cancer, all primary end points of safety and efficacy of alpha1-oleate treatment are reached, as evaluated in an interim analysis. Intra-vesical instillations of alpha1-oleate triggers massive shedding of tumor cells and the tumor size is reduced but no drug-related side effects are detected (primary endpoints). Shed cells contain alpha1-oleate, treated tumors show evidence of apoptosis and the expression of cancer-related genes is inhibited (secondary endpoints). The results are especially encouraging for bladder cancer, where therapeutic failures and high recurrence rates create a great, unmet medical need.
Conflict of interest statement
C.S. holds shares in HAMLET Pharma, as a representative of scientists in the HAMLET group. Patents protecting the use of the alpha1 peptide were filed previously (Biologically active complexes and therapeutic uses thereof; GB 201707715 priority date 14/05/2017, PCT/EP2018/062396 filing date 14/05/2018; inventors: C.S., A.N., J.Ho). No specific patents have been filed based on this study. Other authors declare no competing or conflicts of interests.
Figures
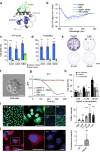

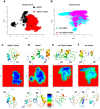

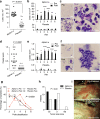


Comment in
-
Uro-Science.J Urol. 2022 Mar;207(3):727-729. doi: 10.1097/JU.0000000000002363. Epub 2021 Dec 14. J Urol. 2022. PMID: 34903031 No abstract available.
References
-
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–674. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

