Hospitalization of Adolescents Aged 12-17 Years with Laboratory-Confirmed COVID-19 - COVID-NET, 14 States, March 1, 2020-April 24, 2021
- PMID: 34111061
- PMCID: PMC8191866
- DOI: 10.15585/mmwr.mm7023e1
Hospitalization of Adolescents Aged 12-17 Years with Laboratory-Confirmed COVID-19 - COVID-NET, 14 States, March 1, 2020-April 24, 2021
Abstract
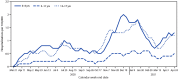
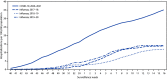
Most COVID-19-associated hospitalizations occur in older adults, but severe disease that requires hospitalization occurs in all age groups, including adolescents aged 12-17 years (1). On May 10, 2021, the Food and Drug Administration expanded the Emergency Use Authorization for Pfizer-BioNTech COVID-19 vaccine to include persons aged 12-15 years, and CDC's Advisory Committee on Immunization Practices recommended it for this age group on May 12, 2021.* Before that time, COVID-19 vaccines had been available only to persons aged ≥16 years. Understanding and describing the epidemiology of COVID-19-associated hospitalizations in adolescents and comparing it with adolescent hospitalizations associated with other vaccine-preventable respiratory viruses, such as influenza, offers evidence of the benefits of expanding the recommended age range for vaccination and provides a baseline and context from which to assess vaccination impact. Using the Coronavirus Disease 2019-Associated Hospitalization Surveillance Network (COVID-NET), CDC examined COVID-19-associated hospitalizations among adolescents aged 12-17 years, including demographic and clinical characteristics of adolescents admitted during January 1-March 31, 2021, and hospitalization rates (hospitalizations per 100,000 persons) among adolescents during March 1, 2020-April 24, 2021. Among 204 adolescents who were likely hospitalized primarily for COVID-19 during January 1-March 31, 2021, 31.4% were admitted to an intensive care unit (ICU), and 4.9% required invasive mechanical ventilation; there were no associated deaths. During March 1, 2020-April 24, 2021, weekly adolescent hospitalization rates peaked at 2.1 per 100,000 in early January 2021, declined to 0.6 in mid-March, and then rose to 1.3 in April. Cumulative COVID-19-associated hospitalization rates during October 1, 2020-April 24, 2021, were 2.5-3.0 times higher than were influenza-associated hospitalization rates from three recent influenza seasons (2017-18, 2018-19, and 2019-20) obtained from the Influenza Hospitalization Surveillance Network (FluSurv-NET). Recent increased COVID-19-associated hospitalization rates in March and April 2021 and the potential for severe disease in adolescents reinforce the importance of continued COVID-19 prevention measures, including vaccination and correct and consistent wearing of masks by persons not yet fully vaccinated or when required by laws, rules, or regulations.†.
Conflict of interest statement
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. Andrea George, Laurie M. Billing, Libby Reeg, Alexander Kohrman, Andrew Weigel, Kenzie Teno, and Jess Shiltz report grant funding from the Council of State and Territorial Epidemiologists. William Schaffner reports personal fees from VBI Vaccines. Evan J. Anderson reports grants for clinical trials from Pfizer, Merck, PaxVax, Micron, Sanofi-Pasteur, Janssen, MedImmune and GSK, and personal fees for consulting from Sanofi-Pasteur, Pfizer, and Medscape, and for data safety monitoring board service from Kentucky Bioprocessing, Inc. and Sanofi-Pasteur; and funding from the National Institutes of Health to conduct clinical trials of Moderna and Janssen COVID-19 vaccines. No other potential conflicts of interest were disclosed.
Figures
References
-
- Shekerdemian LS, Mahmood NR, Wolfe KK, et al.; International COVID-19 PICU Collaborative. Characteristics and outcomes of children with coronavirus disease 2019 (COVID-19) infection admitted to US and Canadian pediatric intensive care units. JAMA Pediatr 2020;174:868–73. 10.1001/jamapediatrics.2020.1948 - DOI - PMC - PubMed
-
- Kim L, Whitaker M, O’Halloran A, et al.; COVID-NET Surveillance Team. Hospitalization rates and characteristics of children aged <18 years hospitalized with laboratory-confirmed COVID-19—COVID-NET, 14 states, March 1–July 25, 2020. MMWR Morb Mortal Wkly Rep 2020;69:1081–8. 10.15585/mmwr.mm6932e3 - DOI - PMC - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

