Rutin prevents tau pathology and neuroinflammation in a mouse model of Alzheimer's disease
- PMID: 34116706
- PMCID: PMC8196535
- DOI: 10.1186/s12974-021-02182-3
Rutin prevents tau pathology and neuroinflammation in a mouse model of Alzheimer's disease
Abstract
Background: Tau pathology is a hallmark of Alzheimer's disease (AD) and other tauopathies. During disease progression, abnormally phosphorylated forms of tau aggregate and accumulate into neurofibrillary tangles, leading to synapse loss, neuroinflammation, and neurodegeneration. Thus, targeting of tau pathology is expected to be a promising strategy for AD treatment.
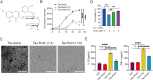
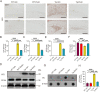
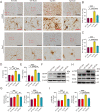
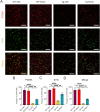
Methods: The effect of rutin on tau aggregation was detected by thioflavin T fluorescence and transmission electron microscope imaging. The effect of rutin on tau oligomer-induced cytotoxicity was assessed by MTT assay. The effect of rutin on tau oligomer-mediated the production of IL-1β and TNF-α in vitro was measured by ELISA. The uptake of extracellular tau by microglia was determined by immunocytochemistry. Six-month-old male Tau-P301S mice were treated with rutin or vehicle by oral administration daily for 30 days. The cognitive performance was determined using the Morris water maze test, Y-maze test, and novel object recognition test. The levels of pathological tau, gliosis, NF-kB activation, proinflammatory cytokines such as IL-1β and TNF-α, and synaptic proteins including synaptophysin and PSD95 in the brains of the mice were evaluated by immunolabeling, immunoblotting, or ELISA.
Results: We showed that rutin, a natural flavonoid glycoside, inhibited tau aggregation and tau oligomer-induced cytotoxicity, lowered the production of proinflammatory cytokines, protected neuronal morphology from toxic tau oligomers, and promoted microglial uptake of extracellular tau oligomers in vitro. When applied to Tau-P301S mouse model of tauopathy, rutin reduced pathological tau levels, regulated tau hyperphosphorylation by increasing PP2A level, suppressed gliosis and neuroinflammation by downregulating NF-kB pathway, prevented microglial synapse engulfment, and rescued synapse loss in mouse brains, resulting in a significant improvement of cognition.
Conclusion: In combination with the previously reported therapeutic effects of rutin on Aβ pathology, rutin is a promising drug candidate for AD treatment based its combinatorial targeting of tau and Aβ.
Keywords: Alzheimer’s disease; Neurofibrillary tangles; Neuroinflammation; Rutin; Synapse loss; Tau pathology.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

