Energy Metabolism of Osteocytes
- PMID: 34117625
- PMCID: PMC8867538
- DOI: 10.1007/s11914-021-00688-6
Energy Metabolism of Osteocytes
Abstract
Purpose of review: In this review, we provide a recent update on bioenergetic pathways in osteocytes and identify potential future areas of research interest. Studies have identified a role for regulation of bone formation and bone resorption through osteocyte mechanosensing and osteocyte secreted factors. Nevertheless, there is a paucity of studies on the bioenergetics and energy metabolism of osteocytes, which are required for the regulation of bone remodeling.
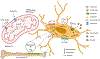
Recent findings: Osteocytes are cells of the osteoblast lineage embedded in bone. The osteocyte lacunocanalicular network within the skeletal matrix is exposed to a unique hypoxic environment. Therefore, the bioenergetic requirements of these cells could differ from other bone cells due to its location in the ossified matrix and its role in bone regulation transduced by mechanical signals. Recent findings highlighted in this review provide some evidence that metabolism of these cells is dependent on their location due to the substrates present in the microenvironment and metabolic cues from stress pathways. Both glycolysis (glucose metabolism) and oxidative phosphorylation (mitochondrial dynamics, ROS generation) affect osteocyte function and viability. In this review, we provide evidence that is currently available about information regarding bioenergetics pathways in osteocytes. We discuss published studies showing a role for hypoxia-driven glucose metabolism in regulating osteocyte bioenergetics. We also provide information on various substrates that osteocytes could utilize to fuel energetic needs, namely pyruvate, amino acids, and fatty acids. This is based on some preliminary experimental evidence that is available in literature. The role of parathyroid hormone PTH and parathryoid hormone-related peptide PTHrP in bone anabolism and resorption, along with regulation of metabolic pathways in the cells of the skeletal niche, needs to be explored further. Mitochondrial metabolism has a role in osteocyte bioenergetics through substrate utilization, location of the osteocyte in the bone cortex, and mitochondrial biogenesis. While there are limitations in studying metabolic flux in traditional cell lines, there are now novel cell lines and sophisticated tools available to study osteocyte bioenergetics to help harness its potential in vivo in the future.
Keywords: Glycolysis; Metabolism; Mitochondria; Osteocytes; Oxidative phosphorylation.
© 2021. The Author(s), under exclusive licence to Springer Science+Business Media, LLC, part of Springer Nature.
Conflict of interest statement
Disclosure Statement: The authors have nothing to disclose
Figures
References
-
- Capulli M, Paone R, and Rucci N, Osteoblast and osteocyte: games without frontiers. Arch Biochem Biophys, 2014. 561: p. 3–12. - PubMed
-
- Buenzli PR and Sims NA, Quantifying the osteocyte network in the human skeleton. Bone, 2015. 75: p. 144–50. - PubMed
-
- Bakker AD, et al., IL-6 alters osteocyte signaling toward osteoblasts but not osteoclasts. J Dent Res, 2014. 93(4): p. 394–9. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

