Deletion of the lactoperoxidase gene causes multisystem inflammation and tumors in mice
- PMID: 34127712
- PMCID: PMC8203638
- DOI: 10.1038/s41598-021-91745-8
Deletion of the lactoperoxidase gene causes multisystem inflammation and tumors in mice
Abstract
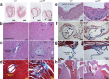
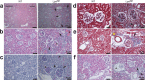
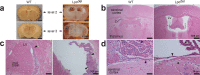
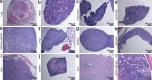
Strongly oxidative H2O2 is biologically important, but if uncontrolled, would lead to tissue injuries. Lactoperoxidase (LPO) catalyzes the redox reaction of reducing highly reactive H2O2 to H2O while oxidizing thiocyanate (SCN-) to relatively tissue-innocuous hypothiocyanite (OSCN-). SCN- is the only known natural, effective reducing-substrate of LPO; humans normally derive SCN- solely from food. While its enzymatic mechanism is understood, the actual biological role of the LPO-SCN- system in mammals remains unestablished. Our group previously showed that this system protected cultured human cells from H2O2-caused injuries, a basis for the hypothesis that general deficiency of such an antioxidative mechanism would lead to multisystem inflammation and tumors. To test this hypothesis, we globally deleted the Lpo gene in mice. The mutant mice exhibited inflammation and lesions in the cardiovascular, respiratory, digestive or excretory systems, neuropathology, and tumors, with high incidence. Thus, this understudied LPO-SCN- system is an essential protective mechanism in vivo.
Conflict of interest statement
The authors declare no competing interests.
Figures




Similar articles
-
Lactoperoxidase-catalyzed oxidation of thiocyanate by hydrogen peroxide: 15N nuclear magnetic resonance and optical spectral studies.Biochemistry. 1991 Jan 8;30(1):118-24. doi: 10.1021/bi00215a018. Biochemistry. 1991. PMID: 1988011
-
Lactoperoxidase-catalyzed oxidation of thiocyanate: polarographic study of the oxidation products.Biochemistry. 1982 Feb 2;21(3):562-7. doi: 10.1021/bi00532a023. Biochemistry. 1982. PMID: 7066307
-
Lactoperoxidase: Properties, Functions, and Potential Applications.Int J Mol Sci. 2025 May 24;26(11):5055. doi: 10.3390/ijms26115055. Int J Mol Sci. 2025. PMID: 40507866 Free PMC article. Review.
-
Lactoperoxidase-catalyzed oxidation of thiocyanate ion: a carbon-13 nuclear magnetic resonance study of the oxidation products.Biochim Biophys Acta. 1992 Oct 20;1159(3):279-85. doi: 10.1016/0167-4838(92)90057-k. Biochim Biophys Acta. 1992. PMID: 1390933
-
Lactoperoxidase as a potential drug target.Expert Opin Ther Targets. 2016;20(4):447-61. doi: 10.1517/14728222.2016.1112378. Epub 2015 Nov 11. Expert Opin Ther Targets. 2016. PMID: 26558497 Review.
Cited by
-
The Effects of 12 Weeks Colostrum Milk Supplementation on the Expression Levels of Pro-Inflammatory Mediators and Metabolic Changes among Older Adults: Findings from the Biomarkers and Untargeted Metabolomic Analysis.Nutrients. 2023 Jul 18;15(14):3184. doi: 10.3390/nu15143184. Nutrients. 2023. PMID: 37513601 Free PMC article.
-
Airway Epithelial Innate Immunity.Front Physiol. 2021 Nov 26;12:749077. doi: 10.3389/fphys.2021.749077. eCollection 2021. Front Physiol. 2021. PMID: 34899381 Free PMC article.
-
Excess iodine exposure acutely increases salivary iodide and antimicrobial hypoiodous acid concentrations in humans.Sci Rep. 2022 Dec 3;12(1):20935. doi: 10.1038/s41598-022-23803-8. Sci Rep. 2022. PMID: 36463312 Free PMC article.
-
Heme Peroxidases at Unperturbed and Inflamed Mucous Surfaces.Antioxidants (Basel). 2021 Nov 12;10(11):1805. doi: 10.3390/antiox10111805. Antioxidants (Basel). 2021. PMID: 34829676 Free PMC article. Review.
-
MicroRNA mediated suppression of airway lactoperoxidase by TGF-β1 and cigarette smoke promotes airway inflammation.J Inflamm (Lond). 2024 Aug 27;21(1):31. doi: 10.1186/s12950-024-00405-x. J Inflamm (Lond). 2024. PMID: 39192275 Free PMC article.
References
-
- Kumar V, Abbas AK, Aster JC. Robbins & Cotran Pathologic Basis of Disease. Elsevier; 2014.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

